
The evolving regulatory policies around biosimilar interchangeability is crucial for driving successful biosimilar adoption in the marketplace.

The evolving regulatory policies around biosimilar interchangeability is crucial for driving successful biosimilar adoption in the marketplace.

Pharmacy errors can happen due to manual processes, but they can be mitigated through adoption of automated IV workflow technology and comprehensive compounding records.

Standardizing observational competencies and utilizing technology to improve compounding accuracy can ensure safety and quality of compounded medications.

Higher costs of efgartigimod alfa are primarily driven by its status as a newly approved, brand-name therapy without a generic alternative.

Food insecurity in rural communities can affect hypertension, but pharmacists can mitigate the impact by educating themselves on nutrition options for low-income individuals.

Firas El Chaer, MD, discusses newly-presented data at ASH demonstrating nuvisertib's effectiveness in myelofibrosis.

Snehal Bhatt discusess the need to individualize antiplatelet therapy decisions based on a comprehensive assessment of each patient's unique risk factors for thrombosis and bleeding.

Obesity is associated with increased distant recurrence and breast cancer mortality in all types of early-stage breast cancer.

Rural pharmacies and hospitals face unique challenges in maintaining adequate staffing and drug supply inventory to serve patients who often travel long distance.
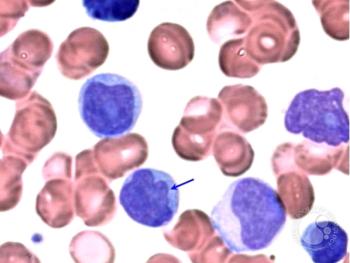
Alison Moskowitz, MD, discusses the clinical considerations, dosing strategies, and the critical role of pharmacists in managing toxicities and tailoring treatment plans for dual-targeted therapy with ruxolitinib and duvelisib.
Emerging research in triple-negative breast cancer highlights the promise of molecular-based approaches to improve treatment outcomes.

AI-powered analysis of mammography images can identify women at high long-term risk of breast cancer, enabling targeted prevention strategies.

Adding anthracyclines to chemotherapy may improve survival in high-risk breast cancer patients.
Advancements in prevention address cancer risk in high-risk patients and BRCA mutation carriers.

Combining CDK4/6 and PDL-1 inhibitors enhances treatment responses in patients with hormone receptor positive and triple-negative breast cancer.

Pharmacists can make a significant impact by providing medication management, promoting nonpharmacologic approaches, supporting patients through life transitions.
Improvements were observed regardless of whether patients initiated ruxolitinib in the second or third line of treatment.

Daily operational tasks and quality assurance measures in pharmacy compounding ensures patient safety and maintains compliance.
Compared with placebo, patients with relapsed/refractory (R/R) follicular lymphoma (FL) who were treated with tafasitamab showed a median progression-free survival (PFS) of 22.4 months.
Ira Zackon, MD, explains where future research lies regarding the implementation of bispecific antibodies in community oncology settings.

The novel camizestrant-ribociclib combination demonstrated encouraging data in advanced, pre-treated ER+/HER2- breast cancer.

Breast cancer prevention vaccines could have significant positive societal impact by reducing cancer diagnoses.
Novel therapies and personalized treatment plans are improving outcomes for patients with ER-positive breast cancer.
Trastuzumab deruxtecan and trastuzumab emtansine address key challenges such as recurrence and metastases in HER2+ early breast cancer treatment.

Pharmacists can help drive the successful adoption of biosimilars by developing educational programs to address provider and patient concerns.

The combination of elacestrant and abemaciclib showed promising 8.7-month progression-free survival in ER+, HER2- advanced breast cancer, including in patients with ESR1 mutations.

Pharmacists are crucial skin care experts and should counsel patients on the importance of adjunct therapies like moisturizers and sunscreen when using certain medications.

Patients with chronic graft-versus-host disease (cGVHD) were most likely to receive belumosudil in the fourth line (33.7%) setting or the fifth through seventh line (33.1%).

The assessment of tumor-infiltrating lymphocytes can provide valuable insights to guide personalized treatment decisions for breast cancer patients.

At the 2024 San Antonio Breast Cancer Symposium, experts discussed the development, clinical impact, and challenges of antibody-drug conjugates (ADCs).