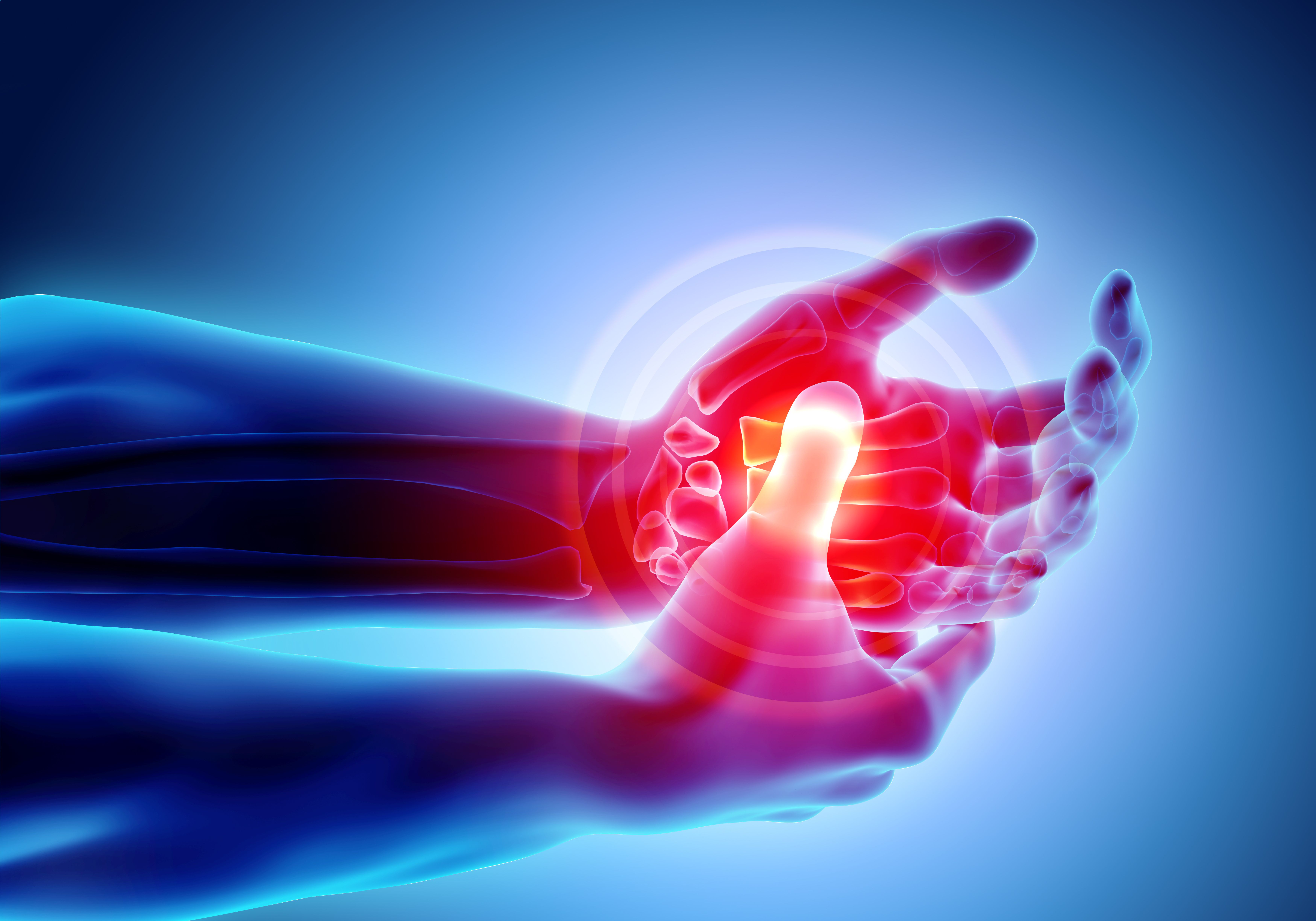
Rheumatoid Arthritis
Latest News

Latest Videos

CME Content
More News

This is the first clinical evidence to show that stimulating the programmed cell death protein 1 (PD1) inhibitory pathway is beneficial for rheumatologic disease.

It is the second anti-TNFα biosimilar approved for use in the US.

Adalimumab-adbm (Cyltezo Pen; Boehringer Ingelheim) is approved by the FDA as an interchangeable biosimilar to Humira.

Individuals who are rituximab-naïve remained stable after 2 years of follow-up, the analysis shows.

Sarilumab is a fully human monoclonal antibody indicated for patients with polymyalgia rheumatica who had an inadequate response to corticosteroids or are unable to tolerate corticosteroid taper.

Research shows that some unsuccessful switches from reference products to biosimilars may be attributed to patient perceptions of reduced efficacy and safety.
YLB113 (Nepexto) produced significantly lower injection site reactions and injection site erythema compared with etanercept in patients with rheumatoid arthritis.

The findings provide an alternative delivery strategy for peptide-based drugs and suggest that such techniques and principles could be applied to a broader range of drugs.

Adalimumab-aacf (Idacio) is indicated for chronic conditions such as rheumatoid arthritis, plaque psoriasis, ulcerative colitis, psoriatic arthritis, hidradenitis suppurativa, and juvenile idiopathic arthritis.

Otilimab also inhibits the biological function of GM-CSF by blocking the interaction of GM-CSF with its cell surface receptor.
Researchers expect to file for FDA approval within the year following the successful phase 3 trial.

Treatment with biologic disease-modifying antirheumatic drugs associated with a higher overall risk, incidence, and recurrence of herpes zoster for patients with rheumatoid arthritis.

Indications for adalimumab include ankylosing spondylitis, Crohn disease, chronic plaque psoriasis, juvenile idiopathic arthritis, moderate to severe rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis.

Some anti-abortion laws may lead to legal action against health care providers who prescribe or provide medications that can be used to induce abortions.
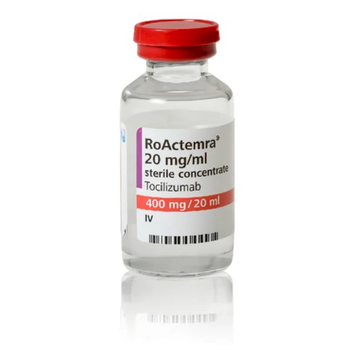
Tocilizumab (Actemra) is indicated for treatment of rheumatoid arthritis, giant cell arteritis, polyarticular juvenile idiopathic arthritis, systemic juvenile idiopathic arthritis, and cytokine release syndrome.
Utilization of the RAPID3 tool can be a beneficial way for pharmacists to complete disease activity assessments without physically seeing the patient in a clinic setting.
Baricitinib (Olumiant) is indicated for the treatment of rheumatoid arthritis and COVID-19 in hospitalized adults.

In addition to the new approval for rheumatoid arthritis, Riabni has already been approved for the treatment of adult patients with non-Hodgkin lymphoma, chronic lymphocytic leukemia, granulomatosis with polyangiitis, and microscopic polyangiitis.

High adherence to treatment is a key first step toward better patient outcomes, including sustained disease control or remission.

Insurers, accountable care organizations, and manufacturers increasingly look for demonstrable clinical outcomes as proof for reimbursement or access to limited distribution networks.
Amidst the frenzy of blockbuster medicines, the connection between the high price of a drug and how well it works is becoming looser.
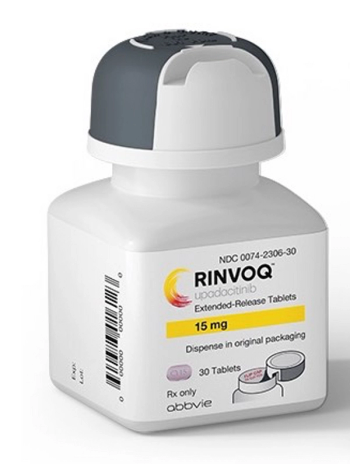
Upadacitinib (Rinvoq) is a Janus kinase (JAK) inhibitor being investigated to treat rheumatoid arthritis, Crohn disease, ulcerative colitis, atopic dermatitis, psoriatic arthritis, axial SpA giant cell arteritis, and Takayasu arteritis
Patients with rheumatoid arthritis taking at least 3 targeted therapies were found to have a greater risk of developing herpes zoster.
Agency greenlights secukinumab’s use for pediatric Treatment of enthesitis-related arthritis and Juvenile psoriatic arthritis.

FDA Approves Abatacept With Calcineurin Inhibitor for Prophylaxis of Acute Graft Versus Host Disease
Abatacept is also approved for adults with moderate to severe rheumatoid arthritis, active psoriatic arthritis, and moderate to severe polyarticular juvenile idiopathic arthritis for children 2 years of age and older.