
Recent economic developments in Europe have impacted the state of specialty pharmacy. Here is an overview of the fluid situation and a comparison with the United States specialty market.

Recent economic developments in Europe have impacted the state of specialty pharmacy. Here is an overview of the fluid situation and a comparison with the United States specialty market.

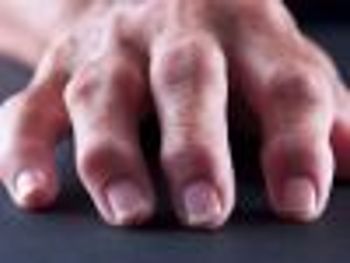
In a retrospective analysis, researchers concluded that rheumatoid arthritis patients had better outcomes when they filled their prescriptions through a mail-order specialty pharmacy rather than a retail pharmacy.

Pfizer's Xeljanz (tofacitinib) is the first Janus kinase inhibitor approved for rheumatoid arthritis and the first oral biologic within the RA class. Dina Rufo of GlobalData discusses the factors that will affect uptake of the drug.
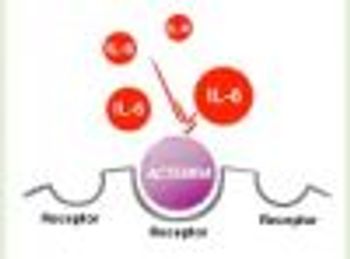
Genentech, Inc, a member of the Roche Group, today announced that the FDA has expanded the approved indication for Actemra (tocilizumab) for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) who have had an inadequate response to one or more disease-modifying antirheumatic drugs (DMARDs).

By the end of this year, it is likely that the FDA will approve more than 20 new specialty medications.
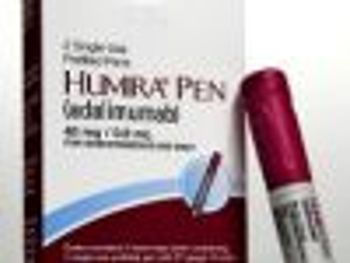
Humira, Abbott's biggest seller, was recently approved by the FDA to treat ulcerative colitis.
When compared with traditional treatments, biologic therapies for RA were found to not be associated with an increased risk of cancer.


A new study finds that patients with autoimmune diseases being treated with immunosuppressive drugs are not at an increased risk of developing shingles.
Phase 3 study findings showed that patients with active moderate to severe rheumatoid arthritis (RA) who received an investigational intravenous (IV) formulation of the anti-tumor necrosis factor (TNF)-alpha therapy Simponi (golimumab) demonstrated significant improvements in signs, symptoms, and disease activity.
The committee voted 8-2 to recommend approval of the investigational agent tofacitinib, and an FDA decision could come as early as August.
Women with autoimmune disorders such as RA generally have fewer children than they would like to have, according to a survey that appeared recently in Arthritis Care and Research.




Findings from a new study show that success in reaching physical goals is associated with reduced arthritis pain and increased quality of life.

