The increase was observed across multiple subgroups, including those with overweight or obesity and hospitalized patients.

The increase was observed across multiple subgroups, including those with overweight or obesity and hospitalized patients.
The drug's safety profile included significant rates of nausea, vomiting, and hematologic toxicities like neutropenia and thrombocytopenia. Although promising, brigimadlin requires careful management of adverse effects, including antiemetic and hematologic support.
Advanced cell therapies are now saving the lives of people whose blood cancer would previously have been incurable. While the UK was quick to recognize the potential of these innovative therapies, patient access to these therapies may change without continued investment in the UK's cell therapy infrastructure.
Glucocorticoids improved outcomes for patients with Kawasaki disease at high risk of IVIG resistance without glucocorticoid-related adverse effects.

Although respondents identified some exciting areas of growth, challenges remain, including the complex insurance system and ensuring patient medication adherence.
Current guidelines recommend a backbone of SLGT2 therapy in patients with HFpEF who have no contraindications.

Investigators state that health care providers should consider the potential use of 2 doses to increase the effectiveness against influenza B.

Shirish Gadgeel, MD, discusses an integrated analysis of the regional TRUST-I study and global TRUST-II study presented at the European Society for Medical Oncology Congress 2024.

The FDA faces a multitude of challenges in regulating artificial intelligence (AI) and machine learning (ML) medical devices.

Behind The Script is a photo submission campaign designed to feature real pharmacists with candid, HIPAA-compliant photos to celebrate American Pharmacist Month.

In this interview for World Standards Week, Nakia Eldridge, PharmD, MPH, of US Pharmacopeia (USP), discusses USP's key achievements in 2024, while outlining the organization's 2025 goals.

The president and CEO of Employers' Forum of Indiana says the pharmacists must be involved and engaged with employers to improve outcomes in health care.

Artificial intelligence (AI) is transforming drug development by automating routine tasks, enhancing clinical trials, and expediting drug discovery, ultimately leading to more personalized treatments.

Offering pharmacy professionals insights into the benefits, importance and challenges associated with switching patients to long-acting injectables to treat schizophrenia.
High-resolution scans can provide a picture of a patient’s brain that typical MRIs cannot, allowing for more detailed insights into the neurological effects of COVID-19.
The trial met its primary endpoint of event-free survival, furthering treatment options for resected, locally advanced head and neck squamous cell carcinoma.
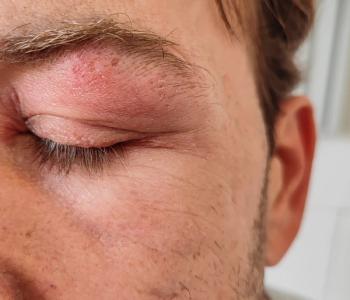
Female and older patients had a greater incidence of herpes zoster ophthalmicus and were more likely to develop ocular complications.

The exemption allows more time for partners to adhere to enhanced distribution security requirements in the Food, Drug, & Cosmetics Act and prevents possible supply chain disruptions.

A single dose RSV vaccine per year can offer optimal protection against 3 full RSV seasons.

Shirish Gadgeel, MD, discusses the promising results of the randomized phase 3 HARMONi-2/AK112-303 study comparing ivonescimab to pembrolizumab in PD-L1–positive advanced non-small cell lung cancer.
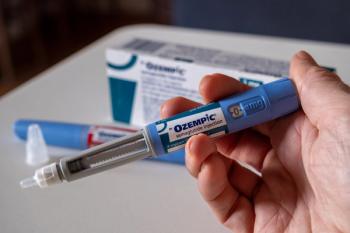
In previous studies, semaglutide was found to modulate dopamine reward signaling and decreased drug rewards (specifically heroin) in rodents.
The real-world study results of ciltacabtagene autoleucel are comparable to data from the CARTITUDE-1 trial, emphasizing its efficacy and safety.

Longer-term data from the phase 3 MARIPOSA trial confirm superior outcomes of a chemotherapy-free amivantamab-vmjw plus lazertinib regimen compared to osimertinib monotherapy as first-line therapy.

The highest pre-term birth rates were consistently observed among those who were Black, American Indian or Alaska Native, or Native Hawaiian or other Pacific islander with public insurance.

Despite its importance, after graduating from pharmacy school, many pharmacists likely experience a decline in confidence in their ability to appropriately evaluate studies and interpret statistical findings.
The discovery marks the first time an antibody has been observed to both assist and block viral infection with SARS-CoV-2.

At the 2024 World Conference on Lung Cancer, Ana Baramidze, MD, PhD, presented 5-year outcomes of the trial, which investigated cemiplimab monotherapy in first-line advanced non–small cell lung cancer (NSCLC) with PD-L1 expression of 50% or greater.
However, the results also showed that vaccination rates were lower in those who are unmedicated for their mental illness and those with substance use disorders.

Pharmacists have developed an expanded role in the team-based approach through medication therapy management.