
Compared with chemotherapy, datopotamab deruxtecan (Dato-DXd) did not achieve a statistically significant overall survival (OS) in patients with inoperable or metastatic HR+/HER2-low or negative breast cancer.

Compared with chemotherapy, datopotamab deruxtecan (Dato-DXd) did not achieve a statistically significant overall survival (OS) in patients with inoperable or metastatic HR+/HER2-low or negative breast cancer.

Pharmacy students discuss a non-traditional learning assignment that used the social media platform, TikTok.

William Carroll, PharmD, network vice president of pharmacy at Hackensack Meridian Health, discusses his hopes for the new partnership.
Further trials are required to examine the long-term feasibility of intravenous immunoglobulin (IVIG) for multisystem inflammatory syndrome.
Approved with 3 new indications, bimekizumab-bkzx is the first and only interleukin-17 inhibitor approved for these diseases.

Ponsegromab displayed significant and robust increases in body weight at the completion of the study for all doses.
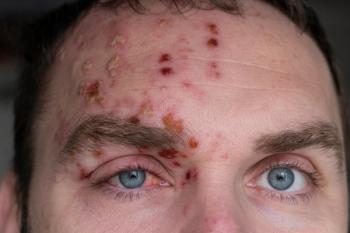
Dermatomal reactions could occur following recombinant zoster vaccine to treat shingles.

The use of devices like Apple watches may offer a more affordable and less burdensome modality to diagnose atrial fibrillation.

Results displayed a non-inferior immune response of the co-administered vaccines, compared to administration in individual visits.
Luspatercept reduced the need for transfusion in patients with myelofibrosis (MF).

Using ultrasound and clinicopathological characteristics, the studied nomograms effectively evaluated recurrence risk in HR-/HER2+ and HR+/HER2+ breast cancer.

Patients with for HER2-positive breast cancer have an increased risk of developing brain tumors.
Though hospitalizations and test positivity remain at modest levels, wastewater analysis indicates high circulation of SARS-CoV-2, especially in western states.

The Drug Supply Chain Security Act (DSCSA) will soon mandate end-to-end tracking of pharmaceuticals throughout the supply chain, with numerous benefits for health systems.

Analyzing consolidation of 8 drug pump libraries into 1 IV infusion library in a 9-hospital system: from standardization to implementation.
The approval is based on positive results from the phase 2/3 IND.227/KEYNOTE-483 trial.
Ten-year follow up data showed a sustained survival benefit with nivolumab-ipilimumab in advanced melanoma.
Peer Reviewed
Implementation of yearly mock code training can reinforce essential knowledge and increase pharmacists’ clinical competence and confidence when faced with actual neonatal and pediatric emergencies.

Pharmacy students discuss the value of collaborative, hands-on learning approaches that leverage technology and social media to enhance learning.
Data from the second interim analysis of the DETECT V trial, presented at ESMO Congress 2024, suggest that a chemotherapy-free regimen combining dual HER2-targeted therapy with endocrine therapy, particularly with the addition of ribociclib, may be an effective treatment for patients.
There is a need for continued research and awareness in the diagnosis and management of alopecia in diverse populations.

With the end of the PREP Act provisions approaching, we want to understand how pharmacists feel about their scopes of practice in each state, and what they think about the future of the profession.

With the introduction of expensive drugs and associated challenges for hospital budgets, pharmacy leaders benefit from strategically managing financial, clinical, and regulatory factors for sustainable care delivery.
Two groundbreaking cell-based gene therapies, exagamglogene autotemcel and lovotibeglogene autotemcel, were recently approved by the FDA for the treatment of sickle cell disease.
Pharmacists’ role in community-based cancer centers are expanding, with a more significant role in treatment plan selection, education, collaboration to create individualized care plans, managing adverse effects, and aiding with financial toxicities.
Overall survival results from the phase 3 KEYNOTE-522 study provide evidence of improved long-term outcomes from this pembrolizumab-based regimen.
Not only do diverse clinical cohorts improve the strength of the data, but it improves public trust in clinicians and the medical system as a whole.
The approval marks the first and only twice yearly, health care professional-administrated injection for both forms of multiple sclerosis (MS).

Researchers suggest prior treatments and follow-up duration may play a larger role.

Disparities continue in younger patients, raising the need for novel interventions to mitigate challenges associated with younger age at diagnosis.