
RSV vaccines were 80% effective in preventing hospitalization, ICU admission, and death among adults aged 60 years and older.

RSV vaccines were 80% effective in preventing hospitalization, ICU admission, and death among adults aged 60 years and older.
Efforts include a mandatory intravenous immunoglobulin ordering form, blood bank gatekeeping, and a stewardship committee to ensure optimized use.
Expanding operations to provide specialty infusions can provide benefits to both patients and oncology practices.
Opioid stewardship pharmacist Maureen Saphire, PharmD, BCGP, focuses on advocating for appropriate opioid access for patients with cancer and sickle cell disease, promoting safe opioid use, and supporting leadership and strategic planning as keys to successful opioid stewardship.
New technology and innovation may help turn the corner on antibiotic resistance.
Clesrovimab reduced RSV-associated hospitalizations and RSV-associated lower respiratory infection hospitalizations by more than 84% and 90% in preterm and full-term infants, respectively.
Increased bone marrow adiposity is associated with progression of monoclonal gammopathy of undetermined significance (MGUS) to multiple myeloma.
Known to be a leading cause of mortality worldwide, cigarette smoke exposure can make the respiratory tract more susceptible to pneumococcal infection.
RBL is the first FDA-approved microbiota-based live biotherapeutic product for adults.
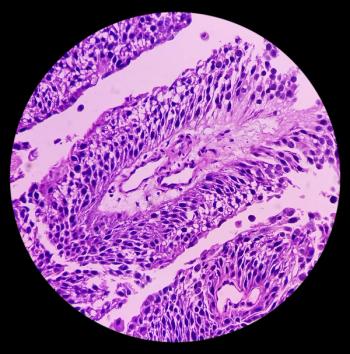
Patients with low-grade intermediate-risk non-muscle-invasive bladder cancer who achieved a complete response rate after 3 months of UGN-102 treatment had an 82.3% duration of response at 12 months.

Insomnia exhibits a dose-response relationship with AF recurrence, indicating that an increased number of insomnia symptoms is associated with heightened probability of AF recurrence.
Educating the public about the dangers of self-medication and the importance of finishing prescribed antibiotic courses plays a crucial role
Jeff Boyer, PharmD, BCPS, discusses his role in opioid stewardship at the Southern Arizona VA Healthcare System, highlighting his work in pain management coordination, interdisciplinary collaboration, patient and provider education, and data-driven strategies to support safe opioid use and address the opioid crisis.

The findings show robust connections between symptoms, with variations in depression scores directly or indirectly influence fatigue and other symptoms.

Less than 16% of high-risk lung cancer individuals have heard or discussed lung cancer screenings with a health care provider.
The novel therapy demonstrated safety and efficacy in a clinical trial.

About 41% and 30% of patients with chronic spontaneous urticaria (CSU), respectively, achieved well-controlled disease status and complete response when treated with dupilumab.

Throughout a new graduate’s career, that provider status will become mainstream.
ELA026 is a first in class antibody therapy targeting signal regulatory proteins.

Julie Sibbesen, PharmD, discussed strategies to address medical misinformation and educate pharmacists on proper sources of information.
The most commonly reported errors included administering the wrong vaccine or administering an expired vaccine.

The randomized, placebo-controlled RESCUE phase 3 trial assessed clofutriben among individuals with ACTH-dependent endogenous Cushing syndrome.
Recently announced trial results demonstrate the efficacy of the biologic in multiple disease indications.

It is more important than ever to have pharmacists and pharmacy professionals practicing at the top of their license to assist patients in pharmacy deserts.

Health system pharmacies can enhance operational and financial performance by leveraging key performance indicators, data-driven tools, and automation, ultimately supporting the broader health system’s adaptability and growth within an evolving health care landscape.

Pharmacists play a critical role in treating patients with breast cancer and can help address their social determinant of health challenges.
In children aged 12, 18, and 24 months, there was no association between maternal COVID-19 exposure and abnormal scores on a neurodevelopment screening.
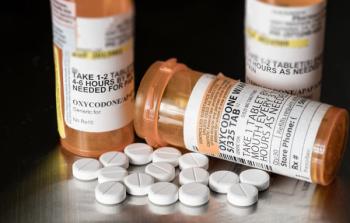
Opioid stewardship coordinator Stephanie Abel, PharmD, BCPS, discusses initiatives such as increasing naloxone access, refining pain management protocols, and addressing stigma around opioid use disorder, highlighting the importance of clear goals and emotional intelligence for effective stewardship programs.

Martha Rumore, PharmD, Esq, discusses the legal and regulatory complexities surrounding tirzepatide compounding following FDA’s removal of the drug from the shortage list, which prompted a lawsuit and led to ongoing uncertainty for 503A and 503B facilities regarding enforcement discretion and future compounding restrictions.

Approximately 15% of patients with polycythemia vera will progress to myelofibrosis.