
Ustekinumab is a human monoclonal antibody targeting the cytokines interleukin (IL)-12 and IL-23, which play a rule in the inflammatory and immune responses.

Ustekinumab is a human monoclonal antibody targeting the cytokines interleukin (IL)-12 and IL-23, which play a rule in the inflammatory and immune responses.

MDL-101 is a proposed novel precision medicine that targets the LAMA1 gene, causing LAMA2 congenital muscular dystrophy type 1a.
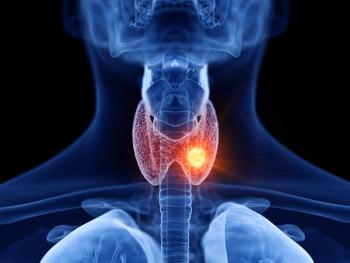
The indication is for adult and pediatric patients aged 2 years and older with advanced or metastatic RET-mutated medullary thyroid cancer (MTC) who require systemic therapy.

Dupilumab is the first biologic medicine for patients with chronic obstructive pulmonary disease to be approved in the US.

Xanomeline and trospium chloride is the first in a new class, offering a new approach with selectively targeting M1 and M4 receptors.

As welcome as this compliance extension may be for those who qualify, it is critical they understand both their current and future regulatory obligations.
The approval marks the only FDA-approved stand-alone therapy for the treatment of Niemann-Pick disease type C (NPC).

The decision is based on positive results from the phase 3 LAURA trial.

Treatment is typically tailored to the individuals, with HCT being the only curative treatment bone marrow failure, but long-term outcomes are generally poorer due to toxicities.
The FDA assigned a Prescription Drug User Fee Act action date of May 22, 2025.

If approved, delgocitinib cream would be the first US treatment indicated for moderate to severe chronic hand eczema.
Approved with 3 new indications, bimekizumab-bkzx is the first and only interleukin-17 inhibitor approved for these diseases.

The decision expands treatment options for patients with transplant-ineligible newly diagnosed multiple myeloma (NDMM).

The rare disease can result in progressive neurological symptoms and organ complications.
Amivatamab-vmjw is a fully-human bispecific antibody targeting EGFR and MET with immune cell-directed activity.
The humanized anti-butyrophilin 3A monoclonal antibody selectively activates Vγ9Vδ2 T-cells.
The approval is based on positive results from the phase 2/3 IND.227/KEYNOTE-483 trial.

Eosinophilic granulomatosis with polyangiitis is a rare and immune-mediate vasculitis that can damage multiple organs and be fatal if left untreated.
The treatment significantly reduced risk of occurrence by 25% compared with endocrine therapy alone.

The non-opioid drug can induce healing of joint cartilage in patients with osteoarthritis.

Lebrikizumab (Ebglyss; Eli and Lilly Company) is a monthly maintenance injection with proven efficacy in adults and children aged 12 to 18 years.
Subcutaneous administration of atezolizumab and hyaluronidase-tqjs had similar efficacy to intravenous administration.

Guselkumab (Tremfya; Johnson & Johnson) is the first and only approved fully human and dual-acting monoclonal antibody blocking interleukin-23 and binding to CD64 receptors.

FCS is a severe, rare genetic disease in which patients have extremely high triglyceride levels, typically above 880 mg/dL.
NS-050/NCNP-03 is being developed to aid individuals with confirmed gene mutations that are treatable with exon 50 skipping therapy.
Full traditional approval follows accelerated approval of the medication last year.

Edaravone and dexborneol sublingual tablets are an innovative first drug to be designed for treatment of stroke and granted this designation.
Previously, the FDA granted fast track designation to ABD-147 (Abdera Therapeutics Inc) for extensive stage small cell lung cancer.
OBX-115 could offer further treatment options for individuals with advanced or metastatic melanoma by enhancing persistence, antitumor activity, and clinical safety of TIL cell therapy.
These companion diagnostics are poised to provide simpler and detailed insights into a patient’s genomic alternations, which could improve outcomes for patients with prostate cancer.