This marks the first and only therapy to receive this designation for Sjögren disease, which currently has no approved treatment.

This marks the first and only therapy to receive this designation for Sjögren disease, which currently has no approved treatment.

Clinical trial analysis results studying VAD-44 in patients with hereditary hemorrhagic telangiectasia (HTT) will be presented at the 2024 American Society of Hematology Annual Meeting & Exposition.
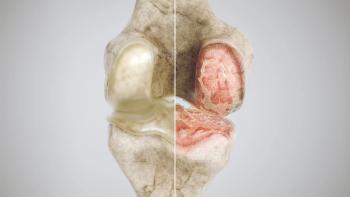
GNSC-001 is a first-in-class gene therapy designed to offer long-term relief of musculoskeletal diseases such as osteoarthritis.

The disorder presents as severe lung disease, with lung transplantation as the only current therapeutic option for long-term survival.

The indication is for adult and pediatric patients 1 year and older with a lysine methyltransferase 2A gene translocation.

The indication is for patients 12 and older with chronic spontaneous urticaria (CSU) whose disease is not adequately controlled with H1 antihistamine treatment.
The tablets become the first and only nilotinib with no mealtime restrictions indicated for this population.
The label includes postmarking reports showing rare instances of pulmonary aspiration for patients undergoing procedures that require general anesthesia or deep sedation.

Eladocagene exuparvovec-tneq is the first FDA-approved gene therapy for individuals with aromatic L-amino acid decarboxylase deficiency.
The designation follows 2 previous orphan drug designations for pancreatic cancer and soft tissue sarcoma.
LBL-034 could be best in class in treating individuals with multiple myeloma.
If accepted, daratumumab would be the first approved treatment for smoldering multiple myeloma.
The regulatory approval corresponds with positive safety and efficacy data from the FELIX trial.

Off-label semaglutide has been linked to serious adverse effects, calling for increased regulation around compounding.

The drug is being investigated for its potential to reduce fibrosis associated with muscular dystrophy that can cause scarring of the heart tissue.

Pharmacy teams are learning to adapt to the challenges of limited distribution networks and orphan drug designation in oncology, with integrated pharmacy support playing a crucial role in enhancing patient access and adherence.

Evidence shows that the agent is not an effective nasal decongestant.

The approval comes after the phase 3 clinical trials MVOR-1 (NCT05296629) and MVOR-2 (NCT05343455).
ELA026 is a first in class antibody therapy targeting signal regulatory proteins.

The randomized, placebo-controlled RESCUE phase 3 trial assessed clofutriben among individuals with ACTH-dependent endogenous Cushing syndrome.

If approved, the combination regimen would be the first FDA-approved treatment for adults with recurrent KRAS mutant low-grade serous ovarian cancer (LGSOC).

If approved, the investigational intravenous gene therapy will be the first disease-modifying therapy for developmental and epileptic encephalopathy (DEE).

HLX14 (Organon) is an investigational biosimilar to denosumab (Prolia/Xgeva; Amgen).
The accelerated approval comes after 48-week major molecular response rate data.

The new indications include pediatric individuals with acute lymphoblastic leukemia and polyarticular juvenile idiopathic arthritis.

The approval marks the first and only abuse-deterrent immediate release oxycodone formulation.
Results demonstrating safety and efficacy in those with anemia in non-dialysis dependent chronic kidney disease and anemia in dialysis-dependent chronic kidney disease were presented at the 2024 American Society of Nephrology Kidney Week.

In postmenopausal women, the gel can reduce moderate to severe hot flashes as well as menopausal changes and symptoms.
Zaltenibart could be an effective treatment option for C3 glomerulopathy as the most proximal inhibitor of the alternative pathway.
Orlynvah is approved for uncomplicated urinary tract infections caused by Escherichia coli, Klebsiella pneumoniae, or Proteus mirabilis.