
Bezisterim, an anti-inflammatory insulin sensitizer, will be authorized to proceed to a phase 2 clinical trial.

Bezisterim, an anti-inflammatory insulin sensitizer, will be authorized to proceed to a phase 2 clinical trial.
Mirdametinib is an oral, allosteric small molecule MEK inhibitor to treat pediatric patients with neurofibromatosis type 1-associated plexiform neurofibromas (NF1-PN).

Previously, the device was indicated for the management of type 1 diabetes in patients aged 2 years and older.

With diagnostic tools, oncology pharmacists and other health care providers can help to identify treatments that have the best probability of working for a specific patient.
The indication is for adults with relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma who were treated with at least 2 prior lines of therapy.

The proposed diagnostic agent has a long half-life, enables imaging the next day, and correlates to a longer shelf life in the pharmacy.
The acceptance will lead to vaccines that provide better protection against the KP.2 strain of SARS-CoV-2.
The FDA assigned the combination a Prescription Drug User Fee Act goal date of April 21, 2025.

The bispecific CAR T-cell therapy for treatment of multiple sclerosis is the first to target both CD19 and CD20.
The breakthrough therapy designation was granted based on ongoing data from the ARTEMIS-001 phase 1 open-label, multi-center trial.
Propofol has a history of chronic shortage due to supply chain constraints.
Vimseltinib (Deciphera Pharmaceuticals) has a Prescription Drug User Fee Act goal date of February 17, 2025.
The combination is the first and only multi-targeted chemotherapy-free regimen that demonstrated superiority compared with osimertinib for non–small cell lung cancer (NSCLC).

The highly potent, selective URAT1 inhibitor may help reduce serum uric acid levels and treat clinically visible tophi.
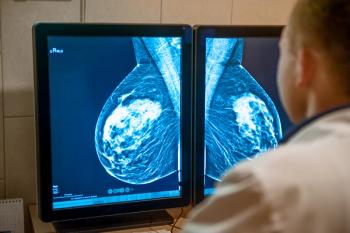
Fam-trastuzumab deruxtecan-nxki has received 4 breakthrough therapy designations, including the latest approval.
Currently, the drug will be studied in the SKYBRIDGE (NCT05652686) trial, a first-in-human, phase 1/2, open-label, dose-escalation and expansion study.

The OTC antibody test can detect current or past infection but should be followed by additional testing to confirm diagnosis.

The hemostatic gel is plant-based and designed to stop and control bleeding within seconds after applied to wound at the point of care.

Durvalumab is a human monoclonal antibody that targets and binds to PD-L1 to interrupt tumor immune-evasion tactics.
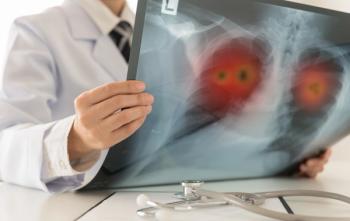
The supplemental biologics license application was granted based on results from the ADRIATIC phase 3 trial among individuals with limited stage small cell lung cancer.

The decision is based positive data from the Deltacel-01 clinical trial.
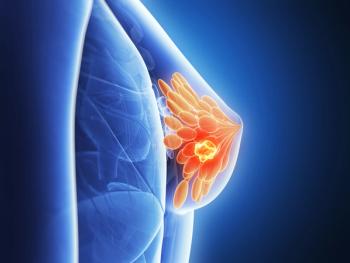
AC699 is being evaluated for patients with estrogen receptor-positive (ER+), human epidermal growth factor receptor 2 negative (HER2–), estrogen receptor 1-mutated advanced or metastatic breast cancer with disease progression on or after at least 1 line of endocrine-based therapy.

The decision is based on clinical findings demonstrating seladelpar’s ability to effectively regulate of alkaline phosphatase values in patients.

The treatment is indicated to improve and maintain visual acuity in individuals with neovascular age-related macular degeneration.
Nemolizumab is the first approved monoclonal antibody specifically inhibiting the signaling of IL-31, which drives disease mechanisms in prurigo nodularis.

The autologous, fully human CD19 chimeric antigen receptor T-cell product, KYV-101, is being studied for the treatment of patients with progressive myasthenia gravis.

The commencement of the phase 1 clinical trial of SAT-3247 is expected in Q3 of 2024.

Previously, an independent advisory panel for the FDA rejected the use of MDMA-assisted therapy for posttraumatic stress disorder (PTSD), citing concerns of the reliability of the clinical trial data.

neffy is an epinephrine nasal spray for the emergency treatment of allergic reactions in adults and pediatric patients.

The FDA also granted rare pediatric disease designation for the drug.