
Craig Beavers and Kyle Fischer debrief on key late-breaking cardiovascular trials from the 2026 American College of Cardiology Scientific Sessions.

Craig Beavers and Kyle Fischer debrief on key late-breaking cardiovascular trials from the 2026 American College of Cardiology Scientific Sessions.

This new podcast will explore therapies, trends, and clinical decisions shaping neurology from a pharmacist's point of view.

Cannabis use is common among patients receiving medications for opioid use disorder and is associated with higher rates of anxiety and other comorbid symptoms.

Oncology pharmacists should weigh lung cancer and diet data, probing pesticides as a possible factor in young nonsmokers.

Combining AI-driven insights with human-first outreach can overcome the emotional and behavioral barriers driving specialty medication nonadherence.

The FDA expands teplizumab-mzwv use to ages 1+ with stage 2 type 1 diabetes, detailing PETITE-T1D safety, EBV/CMV precautions, and pharmacist support to delay onset.
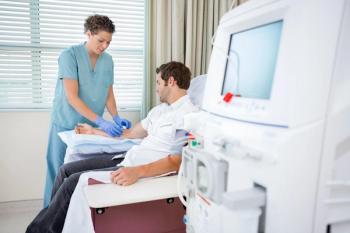
Two studies presented at the World Congress of Nephrology 2026 highlight nonpharmacological strategies that may help preserve kidney function in at-risk populations.
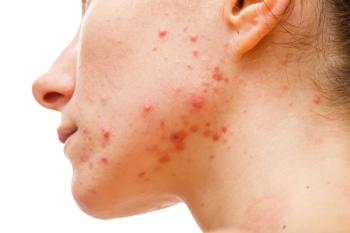
Pharmacists drive acne management by choosing proven OTC ingredients, coaching skin care routines, debunking trends, and knowing when to refer patients to specialists.

The once-weekly Saphnelo Pen offers adult patients with systemic lupus erythematosus a new, at-home alternative to monthly intravenous infusions, supported by robust phase 3 data.
The emerging data suggest a potential adjunctive strategy to target microbial drivers of head and neck squamous cell carcinoma.
Large real-world analysis supports potential renal and survival benefits of urate-lowering therapy in patients with chronic kidney disease and hyperuricemia.

The actions will support the development of psychedelic-based therapies for PTSD, depressive disorders, and substance use disorders.

Sports Pharmacy Summit unites global experts on anti-doping, supplement safety, and data-driven performance to protect athlete health.

The reclassification of medical marijuana to Schedule III marks a major policy shift that expands research and introduces new potential health care challenges.

This week's episode highlights a new executive order signed by President Trump, new expanded FDA approvals, and a conference roundup.

Rapport Therapeutics' RAP-219 achieved seizure freedom in 24% of patients with drug-resistant focal epilepsy over 8 weeks in a phase 2 trial.

A cognitive neurologist outlines the current Alzheimer disease treatment landscape.

How community oncology practices are expanding access to innovative therapies while navigating reimbursement and care delivery challenges.

The treatment's approval was supported by positive evidence from the phase 1/2 CHORD clinical trial.

Emerging data found that initiating low-dose aspirin at giant cell arteritis diagnosis was associated with reduced 1-year major cardiovascular events but an increased risk of major bleeding.


Pharmacists must remain in a constant state of readiness by implementing structured shortage management plans and guiding patients through access challenges as supply disruptions persist.

Extended data from ACHIEVE-4 reinforce the durability of oral GLP-1 receptor agonist therapy, with implications for long-term diabetes management and pharmacist-led care.

Lifileucel treats advanced melanoma by harvesting, expanding, and reinfusing a patient's own tumor-infiltrating immune cells.

New Yale University research offers reassurance for pregnant patients and their clinicians, although evidence of lasting structural damage remains a concern.

Tracking PDUFA decisions and early planning can lead to safe, rapid access to new medications for patients.

AI is delivering immediate clinical value in neurology—from ambient scribes and clinical decision support to trial recruitment and medical education.

Dupilumab becomes the first biologic approved for children aged 2 to 11 years with uncontrolled chronic spontaneous urticaria, marking a significant expansion in treatment options and pharmacist-led care considerations.

A Harvard population health researcher presents findings linking adolescent food insecurity to a cluster of brain health risk factors.