
The VANISH-303 and VANISH-306 studies provide analysis of tablets that treat vulvovaginal candidiasis, which is vaginal yeast infection.

The VANISH-303 and VANISH-306 studies provide analysis of tablets that treat vulvovaginal candidiasis, which is vaginal yeast infection.
An embryonic stem cell model allowed investigators to study the genetic architecture first in hypothalamic progenitor cells, prior to their full development, and then nucleus-like hypothalamic neurons.

Ron Aung-Din, MD, the clinical medical advisor of Pyscheceutical, and Chad Harman, the CEO of Pyscheceutical, discuss a novel approach for administering psychedelics for the treatment of brain disorders that does not induce hallucinogenic effects.

Growth of telehealth means pharmacists can play an important role in patient care even when working from home.
IVIG also has an important therapeutic role in cases with multisystem inflammatory syndrome in children (MIS-C), the researchers said.

These results are based on a case report of a patient with LCV associated to sensitive neuropathy for whom systemic treatment was ineffective.
The investigators said that this analysis is the most comprehensive assessment of vaccination rates among US hospital personnel to date, using data from 3.3 million HCP.

According to the researchers, almost half of the caregivers surveyed responded that the people they are caring for rely on them more than ever before, with 94% of respondents indicating that they put the needs of the person they care for above their own during the COVID-19 pandemic.
Pharmacists have the potential to make a positive impact by screening patients, providing counseling, encouraging beneficial interventions, and helping tailor patient regimens based on personalized needs.

Diagnosis, treatment options, and the role of the pharmacist in patient care are all important factors when looking at multiple sclerosis as a whole.

Ray Tancredi, RPh, MBA, divisional VP of specialty pharmacy development and brand Rx/vaccine purchasing at Walgreens, discusses key drugs in development that are slated to launch in 2022.

Sotrovimab found not inferior to intravenous administration for the early treatment of mild-to-moderate COVID-19 in high-risk, non-hospitalized patients 12 years of age and older.
Non-classical monocytes, a type of innate immune cell that signals molecule to alert other immune cells to threats, could be potential target for COVID-19 therapies.
No differences found in all-cause mortality or stroke between patients with aortic stenosis at intermediate surgical risk who had transcatheter aortic valve replacement or surgery.

Besermi is an interferon alfa-2b indicated for the treatment of adults with polycythemia vera.
Hepatitis A is most likely to be contracted from contaminated food, water, or close contact with a person or object that's infected.

About 29% of the risk for obsessive-compulsive disorder is attributable to differences in single nucleotide polymorphisms between individuals and the control group, study results show.

The model could help researchers and public health officials interpret the significance of existing variants and design tailored public health responses for various scenarios based on a variant’s characteristic.

The cells, named nexus glia, resemble critical glial cells called astrocytes in the brain, and when these cells were removed, heart rate increased.
Physicians advised the individual to adopt the Low Inflammatory Foods Everyday (LIFE) diet, which is a nutrient dense, whole food, plant-based diet.
Galectin-1 may offer a protective function for the kidneys in patients with type 2 diabetes at high risk of diabetic nephropathy.
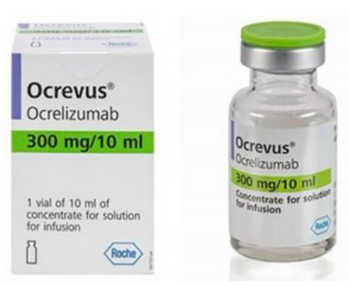
Ocrelizumab is indicated for the treatment of relapsing forms of multiple sclerosis, including clinically isolated syndrome, relapsing-remitting MS, and active secondary progressive disease, in adults.

ADHD is a mental health disorder that includes a combination of persistent challenges, such as difficulty paying attention, hyperactivity, and impulsive behavior.
Within 1 or 2 days of injury, therapeutic B cells release a complex mix of specific pro- and anti-inflammatory molecules, which can affect molecular processing and facilitate healing.
The results showed that one 300 mg dose of AZD7442 reduced the risk of developing symptomatic COVID-19 by 83% and one 600 mg dose reduced the risk of developing severe COVID-19 or death (from any cause) by 88% compared to the placebo.

The FDA has approved maribavir for the treatment of adults and pediatric patients aged 12 years or older and weighing at least 35 kg with post-transplant cytomegalovirus infection/disease that is refractory to treatment with ganciclovir, valganciclovir, cidofovir, or foscarnet.

Investigators have developed an encapsulated and immobilized individual bacterium cell in crystalline polymeric matric that not only kills the bacterium but preserves and stabilizes the dead cell against high temperature, moisture, and organic solvents. The method utilizes metal-organic frameworks (MOFs).

Survey results show 81% think the psychoactive drug will play a role in chronic pain management.

Reports of depression, anxiety and stress have surged since the beginning of the COVID-19 pandemic in 2020.