Lung Cancer
Latest News
Latest Videos
CME Content
More News
Advances in targeted therapies, combination approaches, and novel treatment strategies are shaping the management of drug-resistant lung cancer.
Insights from ESMO 2025 highlight advancements in non-small cell lung cancer treatment, focusing on novel therapies and molecular diagnostics.
TACTI-004 Trial Studying Eftilagimod Alfa With Pembrolizumab in Advanced NSCLC to Begin Evaluation
TACTI-004 follows 2 clinical trials, TACTI-002 and INSIGHT-003, which also assessed efti with pembrolizumab in first-line treatment of non–small cell lung cancer (NSCLC).
Aumolertinib and osimertinib show similar efficacy and safety when treating patients with EGFR-mutant non–small cell lung cancer (NSCLC), providing valuable insights for treatment optimization.
The approval was based on positive efficacy and safety indications from the IMforte clinical trial of patients with extensive-stage small cell lung cancer (SCLC).
Recent findings reveal that stereotactic radiation therapy (SABR) offers long-term survival outcomes comparable to surgery in patients with early non–small cell lung cancer (NSCLC).
Interim trial data presented at the 2025 World Conference on Lung Cancer shows pumitamig’s promising efficacy in patients with extensive stage small cell lung cancer (ES-SCLC).

FDA Approves Keytruda Qlex Subcutaneous Injection for Adult and Pediatric Patients With Solid Tumors
The approval is supported by findings from the MK-3475A-D77 study.
NCCN updated its small-cell lung cancer guidelines, introducing lurbinectedin for maintenance therapy and removing the chemotherapy-free interval for better treatment strategies.

Cemiplimab was also superior in improving progression-free survival, objective response rate, and duration of response in patients with non–small cell lung cancer (NSCLC).
Promising phase 2 results for taletrectinib in ROS1+ lung cancer show significant tumor shrinkage and manageable adverse effects.

A recent phase 3 study reveals osimertinib combined with chemotherapy offers unprecedented survival rates for patients with advanced EGFR-mutated lung cancer.
When combined with an anti–PD-L1 therapy, tarlatamab significantly improves overall survival in patients with extensive-stage small cell lung cancer (ES-SCLC).
The treatment was generally well-tolerated in patients with ROS1-positive (ROS1+) non–small cell lung cancer (NSCLC).

Experts discuss radon—the second leading cause of lung cancer.
Joshua K. Sabari, MD, highlights pharmacists’ roles in coordinating access, financial assistance, and early adverse effect management for patients starting zongertinib (Hernexeos).
Joshua K. Sabari, MD, explains zongertinib’s (Hernexeos) favorable safety profile and quality-of-life benefits over intravenous antibody-drug conjugates in patients with HER2-mutated non-small cell lung cancer (NSCLC).

Joshua K. Sabari, MD, discusses how zongertinib’s inclusion in the National Comprehensive Care Network (NCCN) guidelines represents a game-changing second-line treatment for HER2-mutant non-small cell lung cancer (NSCLC).
The FDA designates olomorasib as a breakthrough therapy, enhancing treatment options for advanced non-small cell lung cancer (NSCLC) with KRAS G12C mutations.

Compared with docetaxel, adagrasib shows significant progression-free survival (PFS) benefits in patients with non–small cell lung cancer (NSCLC).
Zongertinib promises significant advancements in patient care.
The action is supported by 3 ongoing clinical trials, which are evaluating izalontamab brengitecan (iza-bren) in non–small cell lung cancer (NSCLC) and other diseases.
The action is significant for patients with extensive-stage small cell lung cancer (SCLC), a population with limited treatment options.
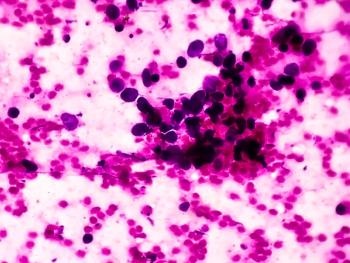
Both preclinical and translational evidence support MET inhibition as a therapeutic approach in patients with small cell lung cancer (SCLC).
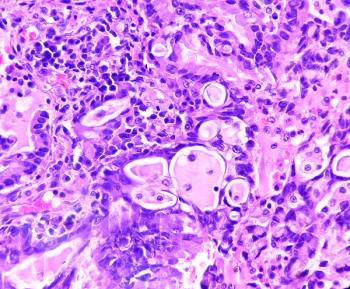
New research links high perilipin 2 expression in lung adenocarcinoma to aggressive disease progression and shorter survival, highlighting potential treatment targets.