
Jeffrey B. Simon, JD, co-chair of the National Opioid Litigation Conference, discusses the historic settlement in a multi-district opioid lawsuit in Texas.

Jeffrey B. Simon, JD, co-chair of the National Opioid Litigation Conference, discusses the historic settlement in a multi-district opioid lawsuit in Texas.
Explain to patients that research Is continually evolving and the need for booster shots does not mean the vaccinations do not work.
Patients treated with upadacitinib achieved clinical remission of ulcerative colitis more frequently through weeks 8 and 52 versus the placebo group.
Agency hopes green light for complex drug device combination improves competition and access more affordable medications.
Nucala is an IL-5 antagonist monoclonal antibody (immunoglobulin G1 kappa) for the add-on maintenance treatment of adult and pediatric patients with severe asthma and with an eosinophilic phenotype.

Zegerid is indicated for the treatment of frequent heartburn.

All patients present opportunities, but patients who might be considered more challenging due to their condition are often the cases where pharmacists might make the greatest difference

Patients may not admit or recognize when they have a brain injury, but pharmacists can be a valuable resource.

The medications may also help tackle the global problem of antibiotic resistance, investigators say.
Investigators call for routine monitoring of vaccination effectiveness as the pandemic continues to evolve.

Corium’s Adlarity system lowers the likelihood of adverse gastrointestinal (GI) events that are normally associated with the oral medication.
The KEYLYNK-010 trial was evaluating a combination of pembrolizumab (Keytruda) and olaparib (Lynparza) in patients with metastatic castration-resistant prostate cancer who progressed following chemotherapy and either abiraterone acetate (Zytiga) or enzalutamide (Xtandi).

This month's generic product news features lanreotide injections, glycopyrrolate oral solution, and vasopressin.

Pfizer Inc and BioNTech seek approval for an additional fourth booster dose of their COVID-19 vaccine for adults 65 years of age and older who received an initial booster of any of the authorized or approved COVID-19 vaccines.
Galcanezumab-gnlm (Emgality) is indicated in adults for the preventive treatment of migraine and treatment of episodic cluster headache.

Co Q-10 helps to support a healthy heart and cardiovascular system.

Pharmacy Times spoke with students at the Idaho State University College of Pharmacy and its partner program at the University of Alaska Anchorage College of Health.
The median survival time of patients was 12 months compared with 7 in the control group, the analysis demonstrates.

Analysis assessing data from the world’s largest twin registry also finds evidence that genetic factors for strong headaches may differ between men and women.

The research team used the National Cancer Institute’s Surveillance, Epidemiology, and End Results database, which provides nationwide information on cancer statistics.
Ramucirumab (Cyramza; Eli Lilly) is FDA-approved for 5 unique cancer indications.
The global impact of GBS causes approximately 150,000 deaths of babies each year.
Moderna is currently evaluating 2 HIV vaccine strategies that involve germline targeting and immune-focusing approaches.

Sleeplessness Is prevalent, and many Individuals struggle to find effective relief and treatment.

BRACAnalysis CDx assay is indicated for use as a companion diagnostic to select patients with germline BRCA-mutated, HER2-negative, high-risk early breast cancer who may derive clinical benefit from olaparib.

Mark Garofoli, director of Experiential Learning Program at WVU School of Pharmacy, discusses the role and impact of pharmacy technicians on opioids in the pharmacy.
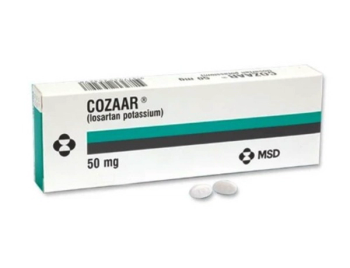
Cozaar is indicated for the treatment of hypertension in adults and children over 6 years of age.
Nexium 24 HR offers relief of frequent heartburns.

Carlie Traylor, PharmD, discussed the Community Pharmacy Fellowship from the National Community Pharmacists Association and CPESN.

It is essential that women are informed of the risks of potentially teratogenic medications because most birth defects are likely to happen early in pregnancy before women become aware they are pregnant.