
OR-449 is being developed for both adult and pediatric patients with adrenocortical carcinoma, as well as other cancers known to express a high level of steroidogenic factor-1.

OR-449 is being developed for both adult and pediatric patients with adrenocortical carcinoma, as well as other cancers known to express a high level of steroidogenic factor-1.

Zilucoplan is among highlighted products of the month.

The impact of RSV includes serious financial challenges, strained relationships, and mental health concerns.
Researchers noted no significant changes in anxiety or depression between patients who were positive or negative for amyloid deposition.
CAR T cell gel eliminated the residual tumor cells in almost all mouse models, allowing the mice to survive when they otherwise would have succumbed to tumor recurrence.

Analysis includes 11 individuals who sat in ergonomic chairs for 8 hours and only got up at the recommended times.

Matt Gibbs, president of Capital Rx, to discusses alternative career paths for students, including independently owned PBMs.

The device features advancements to improve patient care, eliminating or reducing the risk of blood leakage around valve implants.
Antibodies help neutralize the virus, though the levels decrease at the 6-month mark, data indicate.

Dental opioid prescriptions are associated with risk of addiction and overdose, but most states are enacting policies that limit the duration of prescriptions for acute pain.

Sarah McDonald, cancer survivor and author of The Cancer Channel, discusses her experience battling 2 different unrelated cancers and the value oncology pharmacists could have provided with a greater presence on her care team.

Many patients have baseline risk factors present at diagnosis because of older age and disease-related elements.

Inequities were also apparent between those who saw and did not see neurologists, analysis shows.
Educating patients, posting on social media, and countering misinformation are critical strategies.

Carina Dolan, PharmD, BCOP, senior director for Clinical Oncology and Pharmaceutical Outcomes at Vizient Inc, discusses the highlights and big picture moving forward for hospitals and health-system pharmacies.
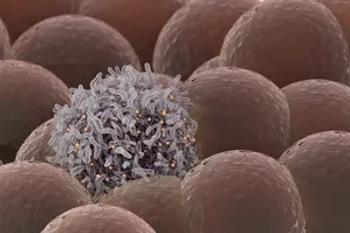
18F-FDG PET/CT detects approximately 73% of tumor lesions, while 68Ga-FAPI finds approximately 94%.

Bruton’s tyrosine kinase inhibitor zanubrutinib (Brukinsa; BeiGene USA, Inc) approved for the treatment of chronic lymphocytic leukemia or small lymphocytic lymphoma.

This is the first indication for tucatinib that extends beyond the treatment of advanced unresectable or metastatic HER2-positive breast cancer.
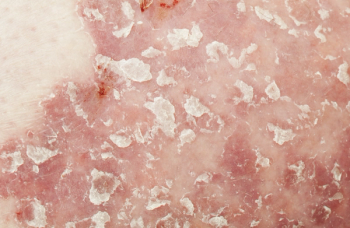
Deucravacitinib is an oral, selective, allosteric tyrosine kinase 2 inhibitor that inhibits cytokine signaling in psoriasis pathogenesis.

Older patients received cancer treatment less frequently during the pandemic, as did patients covered by Medicaid.

November 27, 2023, marks date set by statute for full implementation of Drug Supply Chain Security Act.

Key trends such as consolidation create an opportunity for independent pharmacies with a community-based approach.

Pharmacists can use behavioral and natural tools to assist patients suffering from chronic, daily stressors and other ailments.
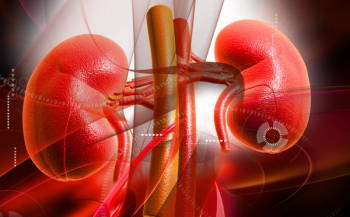
Data from a randomized clinical trial supports previous clinical findings on SGLT2 inhibitors and their ability to slow the progression of chronic kidney disease.

This week's episode features Amina Abubakar, PharmD, AAHIVP, CEO of Avant Pharmacy & Wellness Center and president of Avant Institute, who discusses her experience in the pharmacy industry and the communities she serves.

There is a lack of funding for attention-deficit hyperactivity disorder and coinciding psychological research compared to the funds generated for autism research.
The relationship between metabolic syndrome and some B vitamins, such as vitamin B12, was only observed among participants who took B12 supplements.
The stool-test is the first-of-its-kind to detect CRC in adults aged 45 to 49 years, with a premarket approval submission planned to be sent to the FDA early in 2023.
Soft tissue reconstruction solutions can leverage a patient’s natural healing response.

Research shows that some unsuccessful switches from reference products to biosimilars may be attributed to patient perceptions of reduced efficacy and safety.