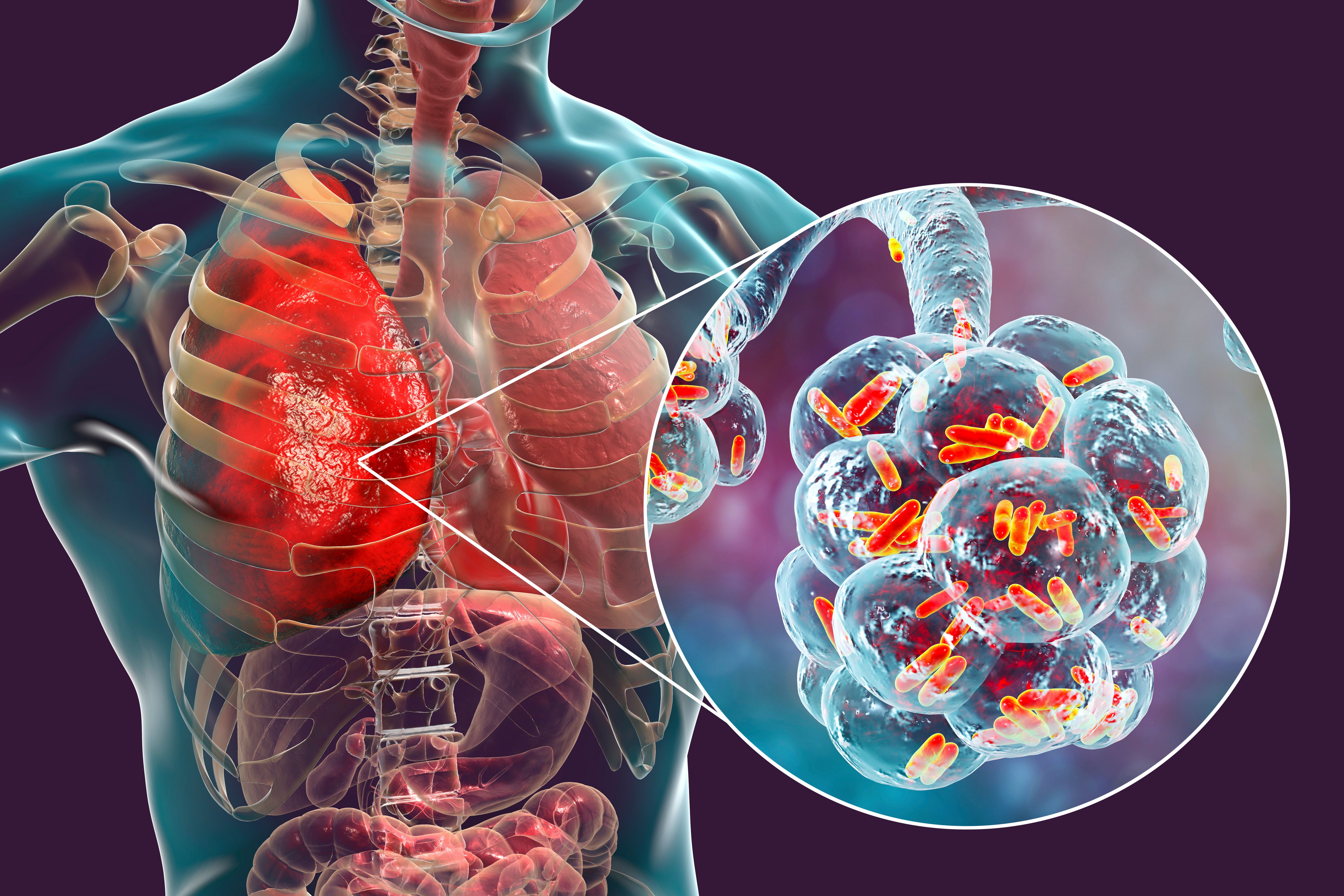
Pneumococcal
Latest News
Latest Videos
CME Content
More News

From newsworthy moments to groundbreaking research, these were the most-read pneumococcal-related articles on Pharmacy Times in 2023.
The study included data on ventilator-associated pneumonia and nonventilator hospital-acquired pneumonia, finding lower rates associated with daily toothbrushing.
Investigators aim to find a substitution for incomplete Freund adjuvant, which is not suitable for use in humans but shows a strong response to antigen-specific systemic immune responses.
If approved, V116 would offer significant improvements in safety and effectiveness for the prevention of pneumococcal disease.
Less than 1% of individuals developed Clostridioides difficile infection (CDI), but for those who previously had a CDI the year before, 12% developed a new infection.

Clinical trial results show the immunogenicity, tolerability, and safety of V116 compared to a pneumococcal 20-valent conjugate vaccine for adults who had not previously received a pneumococcal vaccine.
Encourage patients to locate and safely store their immunization records

Influenza, COVID-19, and respiratory syncytial virus will all peak in the coming months.

According to the investigators, Streptococcus pneumoniae and non-typeable Haemophilus influenzae are dominant pathogens of otitis media after birth.

Use of both nutrition and treatment can help children and elderly populations, especially those in developing countries where vaccine availability is limited.
Serotype 19A is still the most prevalent serotype for pneumococcal disease, with study authors calling for additional studies to determine the full burden of invasive pneumococcal disease.

Pneumococcal serotype 3 was prevalent regardless of vaccination status, with the most common cases being in those with chronic lung disease.

FDA Approves Pentavalent Vaccine for Most Common Serogroups That Cause Meningococcal Disease
Penbraya (Pfizer Inc) is the first and only approved pentavalent vaccine that confers protection against the most common meningococcal serogroups—A, B, C, W-135, and Y, in individuals 10 through 25 years of age.
Study authors suggest that saliva samples can be a complementary testing method to provide additional information on pneumococcal carriage and serotypes that may be undetected by nasopharyngeal testing.
Study data suggest that both vaccines induce similar functional antibody responses against pneumococcal serotype 3.
Study is the first to compare SARS-CoV-2–associated sepsis and presumed bacterial sepsis on mortality and incidence.

Community pharmacies and organizations can collaborate to tailor strategies and interventions and address the challenges and specific needs of the community.

By educating their patients on the provisions of the Inflation Reduction Act, pharmacists can have an even greater impact by increasing vaccine uptake in historically underserved groups.

Senate Bill 219 will also allow pharmacists to independently administer influenza and COVID-19 tests waived by the clinical laboratory improvement amendments.
Best practices to train staff and team members to engage patients and to ease the new vaccine offerings into the workflow are also discussed.

Pharmacists can play a crucial role in educating parents and helping remind them about the childhood vaccine schedule, which can be quite complex.

Investigators from Griffith University stated that technology has helped the development of vaccines that are safe to use and induce strong immune responses against Strep A.

Cost considerations for the rollout of new pneumococcal vaccines are discussed by a panel of medical experts.

Dr Schaffner underscores the significance of multidisciplinary teams in the effort to optimize vaccination rates for pneumococcal disease.

Christina Madison, PharmD, FCCP, AAHIVP, discusses disparities in patient populations directly influencing limited access to appropriate treatment and preventative care.