News
Article
Clinical Overview: Fecal Microbiota Transplantation vs Live Biotherapeutic Products for Management of Recurrent C Difficile Infection
Although antibiotics are the standard-of-care treatment for Clostridioides difficile infection, they can cause further microbiota changes that predispose patients to recurrence.
Clostridioides difficile infection (CDI), the leading cause of antibiotic- and health care-associated infective diarrhea in the United States, is associated with almost half a million infections annually.1,2 Accumulating evidence suggests that dysbiosis, defined as a perturbation in the intestinal microbiota composition characterized by reduced richness and diversity, is implicated in development of and recovery from CDI.3-7
Image credit: Saiful52 - stock.adobe.com
Although antibiotics are the standard-of-care treatment for CDI,8 they can cause further microbiota changes that predispose patients to recurrence. Approximately 20%-35% of patients experience recurrent CDI (rCDI), of whom up to 60% experience subsequent recurrences.9
As prevention and management of rCDI has continued to present a challenge, interest has been re-ignited throughout the past decade in the application of a microbiota-based therapeutic for CDI to aid in microbiome restoration. There is a strong historical basis for such methodologies, as fecally-originated products have been used since the fourth century when Ge Hong used “yellow soup” for diarrheal disease.10
Fecal microbiota transplantation (FMT) from healthy individuals to patients with CDI has been used increasingly over the past several decades to restore healthy flora in the gut, thus breaking the cycle of recurrent CDI.11 Indeed, FMT has emerged as a well-recognized strategy for rCDI management. Further, CDI treatment guidelines published by the Infectious Diseases Society of America and the American College of Gastroenterology now endorse FMT as a recommended therapy for rCDI unresponsive after 2 or more recurrences treated with antibiotics.12,13
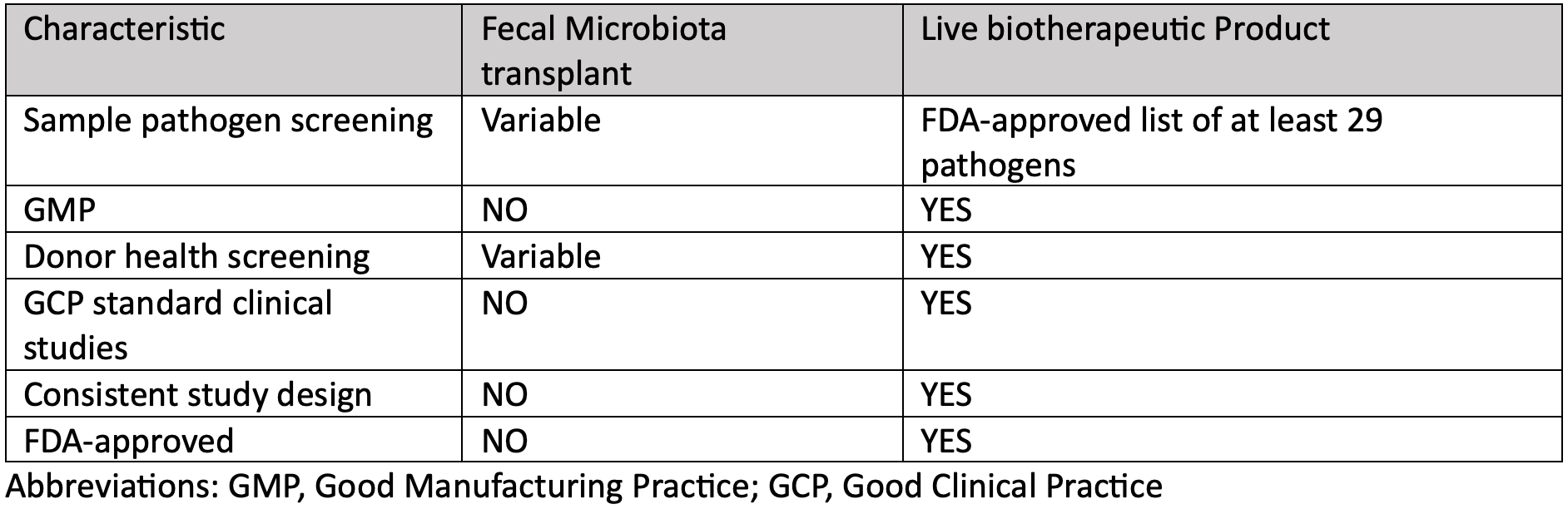
However, FMT is a procedure that is not FDA-approved and has been performed under an investigational new drug (IND) application. Thus, FMT has been notoriously unstandardized with variability reported in fecal material screening and sources, collection procedures, preparation and storage processes, and routes of administration/delivery.14,15
Even the use of centralized manufacturing and storage banks has been noted to carry safety concerns related to the number of patients who may be exposed to a particular donor and particular manufacturing practices. These safety concerns include the transmission of infectious agents to at least 6 patients and ensuing death of 2, which prompted the FDA to issue a warning in 2020 regarding the potential risk of serious or life-threatening infections with the use of FMT.16 Therefore, at this time, the FDA no longer intends to extend enforcement discretion with respect to the IND requirements for distribution of FMT products obtained from stool banks.
The approval of live biotherapeutic products (LBPs) has been a much-needed development in microbiota-based therapies for CDI. Two LBPs are currently FDA-approved for the prevention of CDI recurrence in adults following antibiotic treatment for a rCDI episode: Rebyota (formerly RBX2660; RBL) and Vowst (formerly SER-109; VOS). A third, VE303, is in phase 3 trials. The designation of this new drug class has caused confusion in differentiating between what qualifies as FMT, an LBP, and even a probiotic. We aim here to clarify these differences.
The FDA has designated LBPs as a biological product meeting 3 criteria:17 contains live organisms, such as bacteria; is applicable to the prevention, treatment, or cure of a disease/condition of human beings; and is not a vaccine.
LBPs, such as RBL and VOS, are FDA-approved, manufactured under good manufacturing practice (GMP) methods, are screened thoroughly for pathogens as per the FDA, and both have undergone rigorous clinical trials. These serve as the major differentiating requirements between FMT and LBP.
Likewise, probiotics are considered dietary supplements and are regulated as such. However, each LBP meets the FDA qualifications differently. RBL and VOS are derived from human stool donors18,19 whereas VE303 is derived from clonal cell banks.20 Furthermore, VOS undergoes an ethanol purification step to ensure the final product contains only spores,21 whereas RBL is a whole-stool product.22
Both RBL and VOS are derived from qualified stool donors who undergo rigorous and continuous screening as part of the standardized manufacturing process. Additionally, donor samples are screened for at least 29 pathogens, as per the FDA requirements. Stool donations are collected at the manufacturing site, stored under controlled conditions, and can be traced back to a specific donor, date, and health status at the time of donation.
Clinicians should be aware of the major differences between FMT and LBPs, including RBL and VOS, and not consider them to be the same. FDA-approved LBPs may have differing compositions but are similar, to a large extent, in terms of rigor of manufacturing, safety, and efficacy.
About the Authors
Anne Gonzales-Luna, PharmD, research assistant professor, Department of Pharmacy Practice and Translational Research, University of Houston College of Pharmacy.
Glenn S Tillotson PhD, FIDSA, FCCP, GST Micro LLC.
References
1. Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015 Feb 26;372(9):825-34.
2. Guh AY, Mu Y, Winston LG, et al. Trends in U.S. Burden of Clostridioides difficile Infection and Outcomes. N Engl J Med. 2020 Apr 2;382(14):1320-1330.
3. Zhang L, Dong D, Jiang C, et al. Insight into alteration of gut microbiota in Clostridium difficile infection and asymptomatic C. difficile colonization. Anaerobe. 2015 Aug;34:1-7.
4. Allegretti JR, Kearney S, Li N, et al. Recurrent Clostridium difficile infection associates with distinct bile acid and microbiome profiles. Aliment Pharmacol Ther. 2016 Jun;43(11):1142-53.
5. Seekatz AM, Rao K, Santhosh K, et al. Dynamics of the fecal microbiome in patients with recurrent and nonrecurrent Clostridium difficile infection. Genome Med. 2016 Apr 27;8(1):47.
6. Chang JY, Antonopoulos DA, Kalra A, et al. Decreased diversity of the fecal Microbiome in recurrent Clostridium difficile-associated diarrhea. J Infect Dis. 2008 Feb 1;197(3):435-8.
7. Theriot CM, Koenigsknecht MJ, Carlson PE, Jr., et al. Antibiotic-induced shifts in the mouse gut microbiome and metabolome increase susceptibility to Clostridium difficile infection. Nat Commun. 2014;5:3114.
8. Johnson S, Lavergne V, Skinner AM, et al. Clinical Practice Guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused Update Guidelines on Management of Clostridioides difficile Infection in Adults. Clin Infect Dis. 2021 Sep 7;73(5):e1029-e1044.
9. Cornely OA, Miller MA, Louie TJ, et al. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis. 2012 Aug;55 Suppl 2:S154-61.
10. Zhang F, Luo W, Shi Y, et al. Should we standardize the 1,700-year-old fecal microbiota transplantation? Am J Gastroenterol. 2012 Nov;107(11):1755; author reply p 1755-6.
11. Hoffmann DE, Palumbo FB, Ravel J, et al. A proposed definition of microbiota transplantation for regulatory purposes. Gut Microbes. 2017 May 4;8(3):208-213.
12. McDonald LC, Gerding DN, Johnson S, et al. Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017 Update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin Infect Dis. 2018 Mar 19;66(7):e1-e48.
13. Kelly CR, Fischer M, Allegretti JR, et al. ACG Clinical Guidelines: Prevention, Diagnosis, and Treatment of Clostridioides difficile Infections. Am J Gastroenterol. 2021 Jun 1;116(6):1124-1147.
14. Kelly CR, Kim AM, Laine L, et al. The AGA's Fecal Microbiota Transplantation National Registry: An Important Step Toward Understanding Risks and Benefits of Microbiota Therapeutics. Gastroenterology. 2017 Mar;152(4):681-684.
15. DeFilipp Z, Bloom PP, Torres Soto M, et al. Drug-Resistant E. coli Bacteremia Transmitted by Fecal Microbiota Transplant. N Engl J Med. 2019 Nov 21;381(21):2043-2050.
16. FDA U. Fecal Microbiota for Transplantation: Safety Alert - Risk of Serious Adverse Events Likely Due to Transmission of Pathogenic Organisms 2020 [cited 2023 06 September]. Available from: https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-alert-risk-serious-adverse-events-likely-due-transmission#:~:text=FDA%20has%20been%20notified%20of,or%20STEC%20(four%20patients).
17. FDA U. Early Clinical Trials with Live Biotherapeutic Products: Chemistry, Manufacturing, and Control Information 2016 [cited 2023 06 September]. Available from: https://www.fda.gov/files/vaccines,%20blood%20%26%20biologics/published/Early-Clinical-Trials-With-Live-Biotherapeutic-Products--Chemistry--Manufacturing--and-Control-Information--Guidance-for-Industry.pdf
18. Khanna S, Sims M, Louie TJ, et al. SER-109: An Oral Investigational Microbiome Therapeutic for Patients with Recurrent Clostridioides difficile Infection (rCDI). Antibiotics (Basel). 2022 Sep 10;11(9).
19. Orenstein R, Dubberke ER, Khanna S, et al. Durable reduction of Clostridioides difficile infection recurrence and microbiome restoration after treatment with RBX2660: results from an open-label phase 2 clinical trial. BMC Infect Dis. 2022 Mar 12;22(1):245.
20. Dsouza M, Menon R, Crossette E, et al. Colonization of the live biotherapeutic product VE303 and modulation of the microbiota and metabolites in healthy volunteers. Cell Host Microbe. 2022 Apr 13;30(4):583-598 e8.
21. McChalicher C, Abdulaziz A, Zhou SS, et al. Manufacturing Process of SER-109, a Purified Investigational Microbiome Therapeutic, Reduces Risk of Coronavirus Transmission From Donor Stool. Open Forum Infect Dis. 2022 Sep;9(9):ofac448.
22. REBYOTA [package insert]. Parsippany, NJ: Ferring Pharmaceuticals Inc; 2022.
Newsletter
Stay informed on drug updates, treatment guidelines, and pharmacy practice trends—subscribe to Pharmacy Times for weekly clinical insights.






