Currently, standard first-line chemotherapy with or without immunotherapy for patients with non-small-cell lung cancer (NSCLC) with HER2 mutations has limited efficacy.

Currently, standard first-line chemotherapy with or without immunotherapy for patients with non-small-cell lung cancer (NSCLC) with HER2 mutations has limited efficacy.

Poor sleep quality is directly related to mortality risk before adjusting for lifestyle or health-related factors, and dementia was noticeably linked to mortality risk in older White males.

Cholesin, a hormone found in the gut, may enhance the cholesterol-lowering capabilities of statins.
Fedratinib is an orally available, small molecule inhibitor of JAK-2 approved for treatment of myelofibrosis.
By utilizing a lower dose, patients can avoid potential adverse events and high financial burden.
Eculizumab (Soliris; Alexion), a C5 inhibitor preventing cleavage into C5a and C5b, is widely considered the first-line therapy for high-risk TA-TMA.

Currently, Danon disease has a poor prognosis and does not have any pharmaceutical therapeutics for treatment or management.
As of now, data suggest minimal risk of drug interactions between oral contraceptives and injectable semaglutide.

As American Pharmacists Month begins, Alan Corley talks the importance of pharmacists and the value of the profession personally and to the overall community.

In a phase 1 trial, the nicotine replacement therapy (NRT) demonstrated a higher mean maximum plasma concentration was achieved faster than with an existing inhaled NRT.
In May 2024, the first biosimilars for aflibercept were approved, which included aflibercept-jbvf (Yesafili; Biocon Biologics), known as MYL-1701P.

Pharmacists and health care providers can counsel around vitamin D supplements and natural intake to increase a child’s vitamin D consumption.
Currently, VLS-1488 is being evaluated in a phase 1/2 trial (NCT05902988) in various advanced tumors, including high-grade serous ovarian cancer (HGSOC).
The findings may offer patients treatment options beyond symptom palliation.

Lowering prescription drug costs for patients, saving taxpayers money, helping small businesses level an anti-competitive playing field: all of this is at stake.
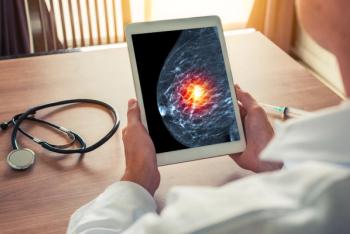
The designation was based on results from the DESTINY-Breast06 trial, evaluating fam-trastuzumab deruxtecan-nxki (Enhertu; AstraZeneca, Daiichi Sankyo) compared with chemotherapy.

The benefits of the daratumumab-based regimen were seen in all patients, including those with high and standard cytogenic risk levels.
The findings support current recommendations for pregnant individuals or those who might become pregnant during the influenza season, specifically those with successive pregnancies.
Positive trial results laid the groundwork for the designation.
Patients exhibited improved disease-free progression with minimal adverse effects.

The weight loss drug continues to show promise in treating a variety of other conditions.
RSV vaccination could be cost-effective among adults 60 years and older, with the ability to decrease vaccine costs and sustain efficacy.

Navenibart was developed to aid treatment of hereditary angioedema (HAE) to provide swift and sustained HAE attack prevention through administration every 3 to 6 months.
Treatment for primary coenzyme Q10 (CoQ10) deficiency can include high-dose oral CoQ10 supplementation, but not all patients respond to this treatment.
Zanubrutinib demonstrated sustained responses in patients with relapsed/refractory (R/R) chronic lymphocytic leukemia or small lymphocytic lymphoma (CLL/SLL).
The results validate the protective benefits of the sodium–glucose cotransporter-2 inhibitors on renal function for those with type 2 diabetes.
A recent study found that indoor allergen exposure, including mouse and cockroach allergens, led to an increased risk of respiratory viral infections in children with asthma.
The accelerated course was safe and effective and did not increase complications in patients with breast cancer post-mastectomy undergoing breast reconstruction.

The AHA acknowledged that complementary and alternative medicines are widely used by heart failure patients but warned that there is limited scientific evidence to support their efficacy.

Ustekinumab is a human monoclonal antibody targeting the cytokines interleukin (IL)-12 and IL-23, which play a rule in the inflammatory and immune responses.