
An upcoming large-scale clinical trial of the Glucose Everyday Matters program will evaluate its safety and efficacy in managing type 2 diabetes.

An upcoming large-scale clinical trial of the Glucose Everyday Matters program will evaluate its safety and efficacy in managing type 2 diabetes.

The upcoming study seeks to analyze whether immature lungs in infants infected with respiratory syncytial virus increases the likelihood of developing asthma later in life.
In addition to overall survival, main efficacy outcomes, such as progression-free survival, objective response rate, and time to response, were compared to chemotherapy alone.
Although antiretroviral therapies are the standard of care treatment for those with HIV, the drugs cannot target latent infections; however, venetoclax, a blood cancer treatment, shows promise.

The fixed-dose abacavir/dolutegravir/lamivudine combination is indicated for the once-daily treatment of children weighing at least 6 kg to <25 kg with HIV-1 infection.
If approved, alectinib would be the only anaplastic lymphoma kinase inhibitor to show reductions in risk of recurrence or death for those with early-stage lung cancer.

Additional research is needed to further evaluate whether adequate emotional support is linked to improvements in health-related quality of life in adults diagnosed with other cancer.
Understanding the virus protein’s insertion mechanisms can be useful in creating new COVID-19 vaccine targets.

AI is revolutionizing pharmacy practice by improving medication management, streamlining workflow, and enhancing patient safety and outcomes.

Manufacturers must find the right balance in drug pricing, so that medications are appealing to both public and private insurers while still remaining profitable throughout their lifecycle.

This year's survey features questions focused on compensation, fulfillment, and burnout.

The risk of developing gestational diabetes was similar between both group prenatal care and traditional individual prenatal care, however, group care may benefit some patients as a treatment option.
Mezigdomide boosted T cell activity and worked in patients with multiple myeloma who were resistant to prior therapeutic agents.

Enhanced utilization of biosimilar products can have significant impact on reducing the overall cost of health care and potentially reducing out-of-pocket costs for patients.

Spatially resolved technology based on next-generation sequencing has the potential to benefit clinical practice and improve the prognosis for cancer patients.
Although the pandemic has negatively influenced the care that patients with cardiovascular conditions receive, the study authors found that there were not significant inequities regarding patients’ race.
Pharmacists play a critical role in educating patients on RSV prevention, how to recognize early symptoms, and when to seek medical help.

As we approach November 27, 2024, new regulations and updates may arise for health system pharmacies to be aware of to remain compliant.
In a previous trial, DARE-19, investigators reported that the SGLT2 inhibitor dapagliflozin did not benefit patients with COVID-19 who had cardiometabolic risk factors.
Heart health may influence long-term brain health as early as middle age.

Pharmacists can play a crucial role in educating parents and helping remind them about the childhood vaccine schedule, which can be quite complex.
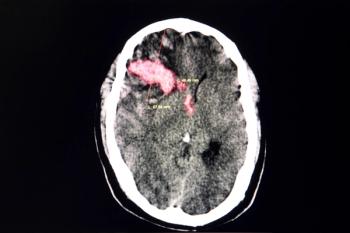
Study finds that statin use is associated with a 12% reduction in the risk of another stroke.

Results from the VALOR-HCM LTE trial at 56 weeks demonstrated that with longer follow-up, mavacamten continued to reduce patient eligibility for invasive septal reduction therapy.
Patients with lymphoma can be immunocompromised due to their disease, anti-cancer therapy, and concomitant immunosuppressive treatments, which make them more vulnerable to developing a COVID-19 infection.

Despite the limited data, prior research indicates there are no serious cannabis-related adverse effects in pediatric patients.

After 1 year of treatment, semaglutide significantly improved physical function, which can improve quality of life outcomes and risk of death.
A combination of statin therapy and antiretroviral therapy may decrease the naturally higher risk of cardiovascular disease in patients with HIV.

When children got less than the recommended 9 hours of sleep or took more than 30 minutes to fall asleep, there was a strong link to impulsive behaviors later in life.

The seamless integration of artificial intelligence and machine learning has the potential to accelerate research and enhance efficiency in a new era of personalized medicine.

Ferric carboxymaltose was not found to have a significant impact on 6-minute walk distance, which is part of the primary outcome’s hierarchical composite of death and hospitalizations from heart failure.