News
Article
Supplements
Vax-Innovation: Incorporating the Newly Approved RSV and Pneumococcal Vaccines Into Community Pharmacy Workflow
Best practices to train staff and team members to engage patients and to ease the new vaccine offerings into the workflow are also discussed.
Recent advancements in the field of vaccines have led to the approval of an additional vaccine against pneumococcal infection and 2 new vaccines against respiratory syncytial virus (RSV). This article provides an overview of these vaccines and outlines engagement strategies and counseling tips for pharmacy professionals to educate patients, involve them in their own care, and help them work toward acceptance and utilization of these novel vaccines. Best practices to train staff and team members to engage patients and to ease the new vaccine offerings into the workflow are also discussed.
anidimi - stock.adobe.com
Pneumococcal 20-Valent Conjugate Vaccine
Prevnar 20, marketed by Pfizer, is a pneumococcal conjugate vaccine that offers expanded coverage against 20 pneumococcal bacteria serotypes, providing enhanced protection against invasive pneumococcal disease.1 Since the approval of Prevnar 20 in June 2021, it is no longer recommended to use Prevnar 13 for older adults (≥ 65 years).2,3 Prevnar 20 is indicated and approved for the prevention of invasive disease caused by Streptococcus pneumoniae in individuals 6 weeks and older and for prevention of otitis media in individuals aged 6 weeks through 5 years.1 In patients 18 years and older, Prevnar 20 is indicated for active immunization for the prevention of pneumonia caused by S pneumoniae. (Please refer to the package labeling for strains included and further information.)
Image From the September 2023 Issue of Immunization Guide for Pharamcists
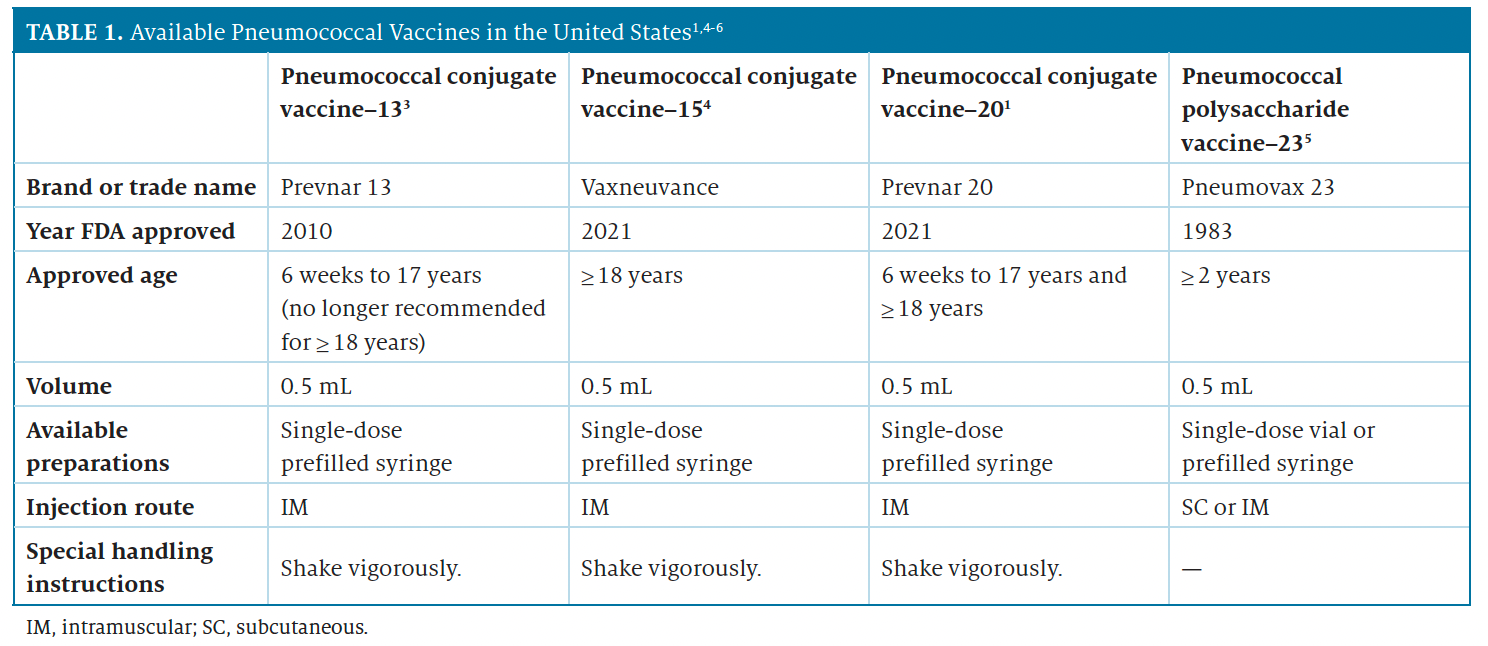
Available pneumococcal vaccines are highlighted in Table 1.1,4-6 In infants and children up to age 17 years, the most commonly reported adverse reactions associated with the use of Prevnar 20 include irritability; pain, redness, and swelling at the injection site; drowsiness; decreased appetite; fever; and headache.1 In adults, the most common adverse reactions were pain at the injection site, muscle pain and fatigue, headache, and arthralgia. The Advisory Committee on Immunization Practices recently updated recommendations to the pneumococcal vaccine series; the schedule can be found on the CDC website (https://www.cdc.gov/vaccines/vpd/pneumo/downloads/pneumovaccine-timing.pdf).2 In the pneumococcal algorithm, if a pharmacy stocked Prevnar 20 for adults, they would meet all “Option A” criteria shown in the CDC table.
RSV Vaccines
RSV is a highly contagious virus that generally circulates seasonally, starting in the fall.7,8 Although it causes infections of the lungs in all age groups, older adults (≥ 60 years), infants, and young children are particularly vulnerable to RSV infection, which can result in lower respiratory tract disease (LRTD), that can lead to pneumonia and bronchiolitis.9 Two vaccines against RSV infection were approved in May 2023.10 The RSV vaccine Abrysvo (Pfizer) and the adjuvanted RSV vaccine Arexvy (GlaxoSmithKline) are indicated for active immunization for the prevention of LRTD caused by RSV in individuals 60 years and older.11,12 The vaccines have been shown to be 85.7% (Arexvy) and 82.6% (Abrysvo) effective at reducing the first episode of RSV-LRTD in individuals with at least 3 symptoms and 66.7% (Arexvy) and 94.6% (Abrysvo) effective in reducing risk of first episode of RSV-LRTD with at least 2 symptoms. The most common adverse reactions are local injection site pain, fatigue, headache, and arthralgia.11,12 In August 2023, Abrysvo was approved for active immunization of pregnantindividuals at 32 through 36 weeks gestational age for the prevention of LRTD and severe LRTD caused by RSV in infants from birth through 6 months of age.13
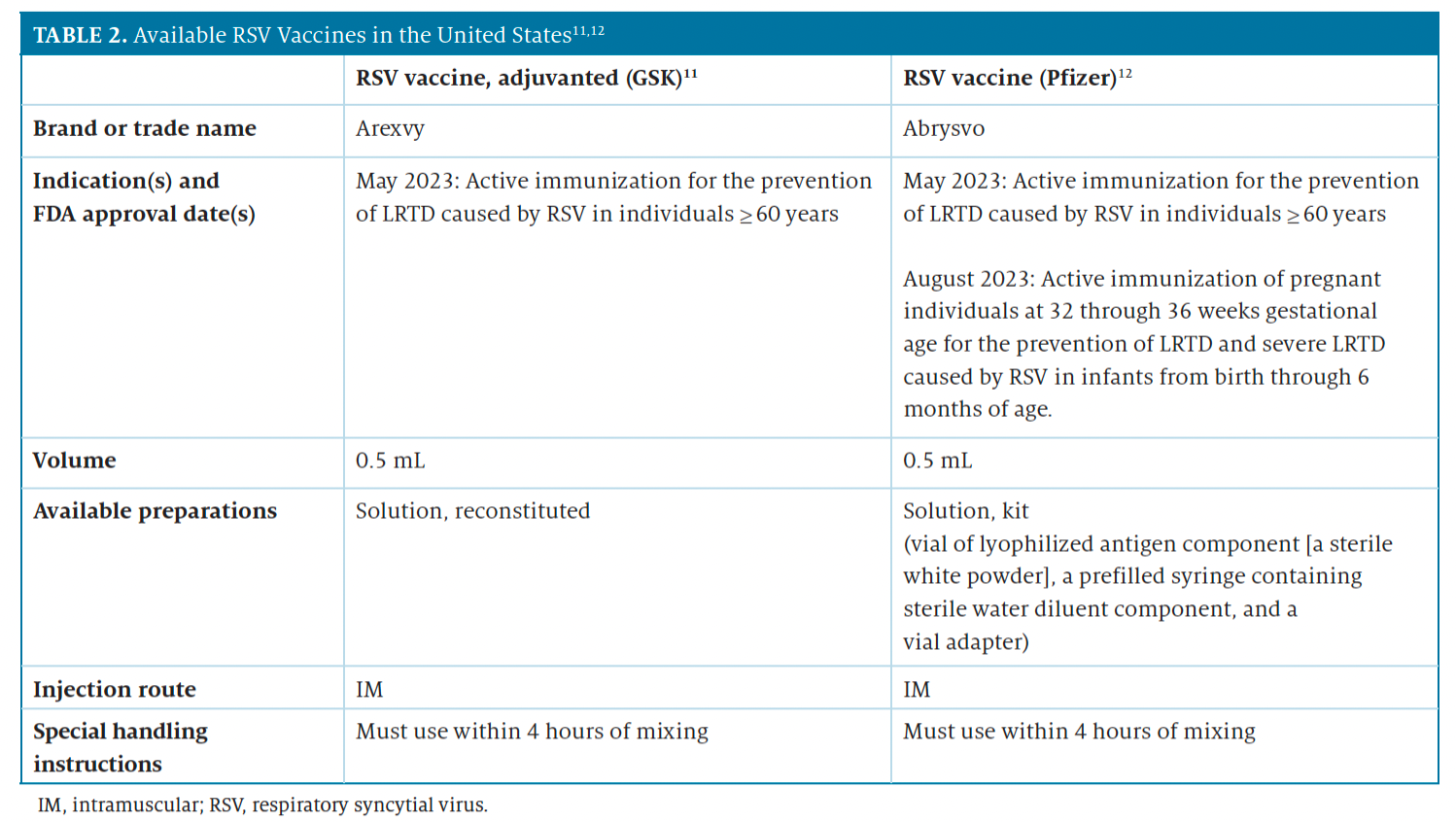
Table 2 describes the 2 RSV vaccines, which are expected to be available in fall 2023.11-12 Both vaccines require reconstitution. For health care providers, knowing which patients would benefit from RSV vaccines and educating them about the advantages of vaccination is critical. For example, older adults with underlying lung disease or a weakened immune system are at greater risk for hospitalizations related to infection; therefore, health care providers should consider recommending the RSV vaccine at the same time they recommend the influenza vaccine.14
Image From the September 2023 Issue of Immunization Guide for Pharamcists
Engagement Strategies and Counseling Tips to Promote Acceptance of New Vaccines Among Patients
Vaccine hesitancy poses a considerable challenge to achieving optimal vaccination rates; this challenge can be further exacerbated with the introduction of new vaccines as patients become aware of new information, policies, and reports of associated risks that can be difficult to understand or, in some cases, misleading or inaccurate.15,16 Pharmacists, as accessible health care providers, are uniquely positioned to engage with patients and address their concerns, ultimately promoting acceptance of these vaccines. The results of a study published in 2022 found that nearly 90% of Americans live within 5 miles of a community pharmacy.17 In addition, results of further studies have shown that patients visit their community pharmacy approximately twice as frequently as they visit primary care offices.18 Some studies have found that patients frequent their pharmacy approximately 35 times per year.19 Accordingly, pharmacists’ relationships with their patients strategically position them to assist patients in accepting new vaccines and/or overcoming vaccine hesitancy. When informing patients about new vaccines, the mnemonic “T.E.A.C.H.” may be beneficial as an engagement tool:
- Trust: Establish a trusting relationship. Building a trusting relationship with patients is essential to promoting acceptance of new vaccines. Leverage your relationship with patients to engage in an empathetic and nonjudgmental conversation. As a health care professional, a pharmacist can help strengthen a patient’s trust in vaccines, thereby helping to prevent them from contracting infectious diseases.
- Educate: Tailor communication and provide clear information. Recognize that each patient has different information needs and communication preferences. When communicating with a patient, make sure you adapt your communication style and language to suit the individual patient’s needs. Ensure they fully comprehend the benefits and risks associated with the new vaccine by using open-ended questions. Make sure you offer evidence-based information about the new vaccine, emphasizing its role in preventing disease and safeguarding public health. When discussing vaccines with the patients, including newly-approved vaccines, capitalize on your relationship with the patient to blend personalized experience with your recommendation. For example, if you know that an older adult patient (≥ 60 years) has frequent lower respiratory tract infections resulting in hospitalizations, tailor the conversation to recommend one of the RSV vaccines. Be certain that the information you’re providing improves patients’ understanding and engagement.
- Address: Vaccine misinformation. Many patients may have misconceptions or may have encountered misinformation regarding new vaccines. Pharmacists serve as an accessible vaccine resource and should be prepared to address these concerns. Offering accurate information from reliable sources such as the CDC and the World Health Organization and debunking common vaccine myths can help ensure patients are making well-informed decisions when encountering new vaccine offerings. When offering information that may be new to a person, consider the concepts of motivational interviewing.20 For example, if a patient is doing research on vaccines, commend their research efforts, and then ask for permission to share new information. Some common motivational interviewing techniques can be found in Figure 1.21,22
Image From the September 2023 Issue of Immunization Guide for Pharamcists

- Counsel: Discuss the vaccine risks vs benefits. Discuss the potential adverse reactions of new vaccines and emphasize the importance of reporting any adverse reactions to the pharmacy. Addressing adverse reactions proactively can help patients feel informed and prepared, reducing vaccine hesitancy. One counseling tip that could be shared is that everything a person does has risks and benefits. For example, you could say, "When you decided to drive your car to the pharmacy today, there was a benefit (getting to a place to purchase your medicine) and a risk (potential car accident). To minimize the risk, you wore a seat belt. The benefits of this vaccine are ___, and the risks are ___. To minimize the risks, we encourage you to ___." This can empower the patient to make an informed decision about their health.
- Highlight: Vaccine safety and development process. Emphasize the rigorous processes involved in vaccine development, including clinical trials and regulatory approvals. Reassure patients about the extensivemonitoring systems in place to ensure vaccine safety post-licensing.
Best Practice Tips for Engaging Team Members, Easing New Vaccine Offerings into the Workflow, and Engaging Patients
With the introduction of new vaccines, it is imperative for pharmacy professionals to implement effective strategies for training and engaging their staff. Integration of new vaccine offerings into the pharmacy workflow effectively requires careful planning, training, and collaboration. Engaging the entire pharmacy team is a vital component to successful integration into the workflow and patient uptake. Following a few best practices can ensure a smooth transition and put your team in an optimal position to educate and engage the patient.
Assess the Team's Needs and Current Level of Training
Successfully introducing a new vaccine requires conducting a needs assessment of your pharmacy team. The assessment should help to identify knowledge gaps and training requirements. Implementing the following best practices during the training process may be helpful.
- Tailor training programs: Develop training programs that are specific to the needs of your team. Consider factors such as experience level, familiarity with vaccine protocols and regulatory guidelines, and everyone’s role on the team.
- Provide up-to-date resources: Ensure the team has access to credible evidence-based resources such as guidelines from the vaccine manufacturer and the CDC. These resources should be available for your team to review before vaccine rollout and kept as a readily accessible resource.
- Provide practical training: Make sure to include hands-on practical training sessions to reinforce proper vaccination technique. Review storage and handling guidelines for all new vaccines and any specific documentation requirements.
Communication is Key
Effective communication among pharmacy team members, before and after rollout, is vital for successful integration of new vaccine offerings and patient engagement. Meet with the team to ensure everyone is aware of new vaccine offerings. Address team member concerns through open dialogue and provide a platform for team members to ask questions. Team members who are educated on new vaccine offerings will be better equipped to provide accurate information and address patient concerns.
Patient Engagement is Crucial to Promote Vaccine Uptake
Engage patients in person by displaying educational materials about new vaccines in prominent areas of the pharmacy including waiting areas, consultation areas, and vaccination rooms. Providing physical patient handouts and education resources can be a great way to get the patient thinking and talking about new vaccine offerings. The handouts can serve as a visual reminder to the patient after they leave the pharmacy. Pharmacy teams also can use digital platforms, automated phone messages, and texting to disseminate new vaccine information and address common questions and concerns. Digital engagement can provide opportunities for patients to make vaccine appointments at a time that is convenient for them.
Patient engagement does not end when the patient receives the vaccine. Following up with the patient given vaccinations can help address any adverse effects or concerns the patient may have. Timely patient follow-up can help to strengthen patient trust in the pharmacy team and minimize vaccine hesitancy in the future.
Conclusions
Pharmacists are strategically positioned as being among the most accessible health care professionals and they have strong relationships with the communities they serve. In the community-based pharmacy setting, pharmacists can work together with colleagues (eg, technicians, interns) to engage patients in conversations about vaccines, especially in closing vaccine gaps and providing education on newly approved vaccines.
About the Authors
Nicole C. Pezzino, PharmD, BCACP, CDCES, is an associate professor of pharmacy practice in the Wilkes University, Nesbitt School of Pharmacy in Wilkes-Barre, Pennsylvania.
Christopher Svetcov, PharmD, is a pharmacy manager at Wegmans Food Market in Allentown, Pennsylvania.
References
- Prevnar 20. Prescribing Information. Pfizer; 2023. Accessed July 28, 2023. https://www.fda.gov/media/149987/download
- Pneumococcal vaccine timing for adults. CDC. Updated March 15, 2023. Accessed July 1, 2023. https://www.cdc.gov/vaccines/vpd/pneumo/downloads/pneumo-vaccine-timing.pdf
- Prevnar 20 BLA Approval. FDA. June 10, 2021. Accessed August 1, 2023. https://www.fda.gov/media/150021/download?attachment
- Prevnar 13. Prescribing Information. Pfizer; 2017. Accessed July 28, 2023. https://www.fda.gov/files/vaccines%2C%20blood%20%26%20biologics/published/Package-Insert------Prevnar-13.pdf
- Vaxneuvance. Prescribing Information. Merck & Co; 2023. Accessed July 28, 2023. https://www.merck.com/product/usa/pi_circulars/v/vaxneuvance/vaxneuvance_pi.pdf
- Pneumovax-23. Prescribing Information. Merck & Co; 2023. Accessed July 28, 2023. https://www.fda.gov/media/80547/download
- Rose EB, Wheatley A, Langley G, Gerber S, Haynes A. Respiratory syncytial virus seasonality - United States, 2014-2017. MMWR Morb Mortal Wkly Rep. 2018;67(2):71-76. doi:10.15585/mmwr.mm6702a4
- Hamid S, Winn A, Parikh R, et al. Seasonality of respiratory syncytial virus - United States, 2017-2023. MMWR Morb Mortal Wkly Rep. 2023;72(14):355-361. doi:10.15585/mmwr.mm7214a1
- Respiratory syncytial virus infection (RSV). CDC. Updated October 8, 2022. Accessed August 1, 2023. https://www.cdc.gov/rsv/index.html
- Melgar M, Britton A, Roper LE, et al. Use of respiratory syncytial virus vaccines in older adults: recommendations of the Advisory Committee on Immunization Practices - United States, 2023. MMWR Morb Mortal Wkly Rep. 2023;72(29):793-801. doi:10.15585/mmwr.mm7229a4
- Abrexvy. Prescribing Information. GSK; 2023. Accessed July 28, 2023. https://www.fda.gov/media/167805/download
- Abrysvo. Prescribing Information. Pfizer; 2023. Accessed August 31, 2023. https://labeling.pfizer.com/ShowLabeling.aspx?id=19589
- BLA approval for respiratory syncytial virus vaccine (Abrysvo). Written communication. FDA. August 21, 2023. Accessed August 31, 2023. https://www.fda.gov/media/171492/download
- RSV in older adults and adults with chronic medical conditions. CDC. Updated July 14, 2023. Accessed August 1, 2023. https://www.cdc.gov/rsv/high-risk/older-adults.html
- Ten threats to global health in 2019. World Health Organization. January 10, 2019. Accessed July 3, 2023. https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019
- Larson HJ, Gakidou E, Murray CJL. The vaccine-hesitant moment. N Engl J Med. 2022;387(1):58-65. doi:10.1056/NEJMra2106441
- Berenbrok LA, Tang S, Gabriel N, et al. Access to community pharmacies: a nationwide geographic information systems cross-sectional analysis. J Am Pharm Assoc (2003). 2022;62(6):1816-1822.e2. doi:10.1016/j.japh.2022.07.003
- Berenbrok LA, Gabriel N, Coley KC, et al. Evaluation of frequency of encounters with primary care physicians vs visits to community pharmacies among Medicare beneficiaries. JAMA Network Open. 2020; 3(7):e209132. doi:10.1001/jamanetworkopen.2020.9132
- Valliant SN, Burbage SC, Pathak S, Urick BY. Pharmacists as accessible health care providers: quantifying the opportunity. J Manag Care Spec Pharm. 2022;28(1):85-90. doi:10.18553/jmcp.2022.28.1.85
- Excellence in motivational interviewing. Understanding motivational interviewing. Motivational Interviewing Network of Trainers. Accessed July 1, 2023. https://motivationalinterviewing.org/understandingmotivational-interviewing
- Zolezzi M, Paravattil B, El-Gaili T. Using motivational interviewing techniques to inform decision-making for COVID-19 vaccination. Int J Clin Pharm. 2021;43(6):1728-1734. doi:10.1007/s11096-021-01334-y
- Talking with patients about COVID-19 vaccination. CDC. Updated November 3, 2021. Accessed July 10, 2023. https://www.cdc.gov/vaccines/covid-19/hcp/engaging-patients.htm

Newsletter
Stay informed on drug updates, treatment guidelines, and pharmacy practice trends—subscribe to Pharmacy Times for weekly clinical insights.