Publication
Article
Supplements
Influenza Activity and Vaccine Effectiveness During the 2021-2022 Season
Author(s):
Influenza viruses typically circulate in the Northern Hemisphere during the winter and spring, but they occasionally can circulate more during the summer months, as did the 2009 H1N1 pandemic strain.
In late 2019, the novel coronavirus known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spread across the globe, causing the disease known as COVID-19.1 A pandemic was declared by the World Health Organization (WHO) in March 2020.2 The CDC speculated that the mitigation measures (eg, stay-at-home orders, face masks, social distancing, hand cleaning protocols, improved building ventilation, and sharp travel declines) put in place to combat COVID-19 would lead to both a reduction in cases and a global decline in the number of influenza cases during the 2020- 2021 season.3
Influenza Activiry
Influenza viruses typically circulate in the Northern Hemisphere during the winter and spring, but they occasionally can circulate more during the summer months, as did the 2009 H1N1 pandemic strain.4,5 So far, SARS-CoV-2 has been able to circulate and surge at different times of the year, primarily because new, more contagious variants have been introduced.6 During the 2020-2021 influenza season, 0.2% of respiratory samples were positive for influenza, whereas positivity reached a peak of 26% to 30% during the prior 3 influenza seasons.3 During the 2021-2022 season, influenza activity had a bimodal pattern, with a positivity slightly above 6%noted in December 2021; this fell to approximately 2% in January 2022 and peaked again at week 15 of 2022 with 10% positivity.7 The majority of cases were attributed to type A (H3N2) influenza.
Types of Influenza Viruses
There are 2 main lineages of human influenza viruses, types A and B.8 For type A, viruses are subdivided by their major antigenic markers, hemagglutinin and neuraminidase. The currently circulating type A strains include H3N2 (accounting for > 99% of cases), which has been circulating since its introduction in the United States during the 1968 pandemic, and (H1N1)pdm09, the strain from the 2009 pandemic.7,9 Other viral characteristics that determine vaccine composition each year include epidemiologic data (ie, particular influenza viruses that circulate around the world at different locations and times) and genetic and antigenic changes.10 The year-to-year genetic mutations that allow influenza to partially evade our antibodies are called antigenic drift.11 This differs from antigenic shift, which refers to a major change in viral hemagglutinin or neuraminidase proteins that may lead to lack of immunity among a population and, ultimately, a pandemic.
Influenza Vaccines and Vaccination
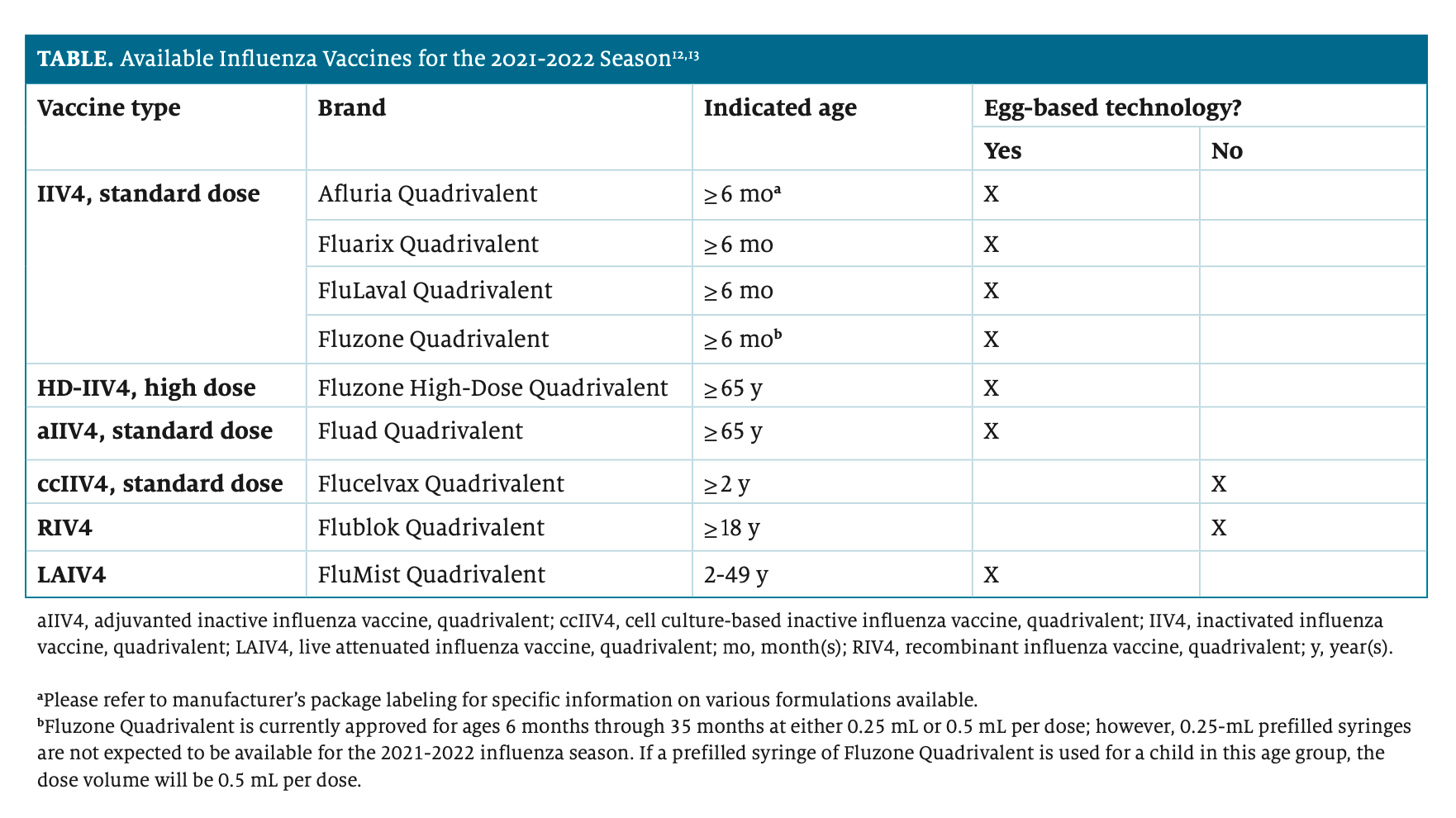
Nine influenza vaccines—1 live-attenuated and 8 inactivated—were available during the 2021-2022 influenza season (Table).12,13 The FDA licenses influenza vaccines for use in individuals at least 6 months of age; recommendations by the CDC’s Advisory Committee on Immunization Practices are in line with this age limit.
All influenza vaccines marketed in the United States are quadrivalent, meaning that they target a type A(H3N2) virus, a type A(H1N1) virus, a type B (Yamagata lineage) virus, and a type B (Victoria lineage) virus.8 The vaccines can be further described based upon whether they are manufactured using egg-based technology (Table).12,13 Egg-based vaccines have been available for the longest amount of time; however, egg adaptation that may occur during the vaccine manufacturing process can result in a vaccine strain not as well matched to the wild-type circulating strain.14,15
Newer vaccine development methods involve using cell-culture or recombinant technology to avoid egg adaptation.14,15 The 2 non-egg–based vaccines available are the quadrivalent cell culture-based inactivated influenza vaccine (ccIIV) Flucelvax Quadrivalent and the quadrivalent recombinant influenza vaccine (RIV) Flublok Quadrivalent.12 All influenza vaccines in the United States contain the same major type A and B strains; however, they may differ slightly based on the technology used to manufacture them.13
The following are key changes to the ACIP recommenda- tions for influenza vaccines in the 2021-2022 season13:
- Flucelvax Quadrivalent is now approved for those aged 2 years and older (previously indicated in those aged ≥ 4 years).
- Influenza vaccines can be coadministered with all other vaccines, including COVID-19 vaccines.
- Vaccination can occur as soon as a recommended vaccine is available for women in their third trimester of pregnancy or children needing 2 doses.
- A severe allergic reaction (eg, anaphylaxis) to any ccIIV or RIV or any egg-based vaccine (inactivated influenza vaccine [IIV], live attenuated influenza vaccine [LAIV], or RIV) is a precaution for the use of a quadrivalent IIV or LAIV. A severe allergic reaction to any ccIIV or RIV (including quadrivalent formulations) is a precaution for the use of a quadrivalent ccIIV or RIV.
In March 2022, the FDA voted to approve the 2022-2023 influenza vaccine composition.16 All influenza vaccines available in the United States remain quadrivalent, offering protection against 4 flu viruses.14 Although there was no change from the 2021-2022 season for the H3N2 and B/Yamagata component, the H1N1 and B/Victoria strains were changed for the upcoming 2022-2023 season.13,16
The following strains are to be included in influenza vaccines for the 2022-2023 season16:
- influenza A (H3N2): A/Darwin/9/2021 (H3N2)-like (egg based) or A/Darwin/6/2021 (H3N2)-like (cell or recombinant based);
- influenza A (H1N1): A/Victoria/2570/2019 (H1N1)pdm09-like virus (egg based) or the A/Wisconsin/588/2019 (H1N1)pdm09-like virus (cell or recombinant based);
- influenza B (quadrivalent vaccines): B/Phuket/3073/2013-like virus (B/Yamagata lineage); and
- influenza B (trivalent and quadrivalent): B/Austria/1359417/2021-like virus(B/Victoria lineage).
Almost every person (aged ≥ 6 months) should receive a flu shot by the end of October each year.13 The influenza vaccine is administered intramuscularly or intranasally (as an LAIV), depending upon the specific product formulation and indication.14 Immunization efforts should start in September and October and before the influenza season, which historically peaks in February; substantial activity can continue into May.17 Children who need 2 doses can receive the vaccine as soon as it is available (possibly in July or August); however, because of a concern about a waning immune response during the influenza season, older adults should not receive it early.
Influenza Vaccine Effectiveness
During the COVID-19 pandemic, low influenza activity increased the uncertainty of estimates for the effectiveness of the influenza vaccine.18 Vaccine effectiveness (VE) is determined through observational studies; however, results from such studies often are influenced by various forms of bias that can make it difficult to interpret outcomes. The US Flu Vaccine Effectiveness Network interim estimates were unable to demonstrate a benefit against mild to moderate influenza in the 2021-2022 season for H3N2, that season’s most commonly circulating strain.
Data on protection against severe disease are not available until after the end of the season. According to the CDC, from 2015 to 2020, VE for influenza vaccines ranged from 29% to 48%.19 There is evidence that the high-dose influenza vaccine and adjuvanted influenza vaccine, licensed for individuals aged 65 years and older, led to a better immune and clinical response.20-23 As such, on June 30, 2022, the CDC Director, Rochelle P. Walensky, MD, MPH, adopted the decision memo approving the Advisory Committee on Immunization Practices preferential recommendation to use higher dose or adjuvanted influenza vaccines over standard-dose unadjuvanted influenza vaccines in this patient population.24
Role of Pharmacists in Educating Patients on Influenza Vaccines
During the 2020-2021 influenza season, the CDC reported that pharmacies surpassed doctor’s offices as the most common place where adults received an influenza vaccine.25 Perhaps due to fears of acquiring COVID-19 and influenza, the influenza vaccination rate in adults at least 65 years old increased from 70% (2019-2020 season) to 75% (2020-2021 season). However, the pediatric influenza vaccination rate decreased over this time period.
The CDC produced a toolkit for pharmacists to help increase influenza vaccination rates in patients, especially in those with vaccine hesitancy, and to improve vaccine confidence.26 As the COVID-19 pandemic continues, influenza has not gone away. It will take the efforts of pharmacists and other providers to manage immunization for both diseases this coming autumn.
Editor’s Note: Guidance from the CDC on a preferential recommendation to use higher dose or adjuvanted influenza vaccines over standard-dose unadjuvanted influenza vaccines in patients aged 65 years and older was updated on June 30, 2022; this change is reflected above. Because the publication in which this article appears was finalized prior to this announcement, it does not include the updated recommendation.
About The Author
Jeff Goad, PharmD, MPh, is a professor and associate dean of academic affairs at the Chapman University School of Pharmacy in Irvine, California.
References
1. Coronavirus disease (COVID-19). World Health Organization. May 13, 2021. Accessed May 23, 2022. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/coronavi-rus-disease-covid-19
2. WHO Director-General's opening remarks at the media briefing on COVID-19—11 March 2020. World Health organization. March 11, 2020. Accessed May 23, 2022. https://www.who.int/director-general/speeches/ detail/who-director-general-s-opening-remarks-at-the-media-briefing- on-covid-19---11-march-2020
3. 2020-2021 flu season summary. Centers for Disease Control and Prevention. Updated October 25, 2021. Accessed May 23, 2022. https://www.cdc. gov/flu/season/faq-flu-season-2020-2021.htm#:~:text=What%20was%20the%202020%2D2021%20flu%20season%20like%3F
4. Flu season. Centers for Disease Control and Prevention. Reviewed September 28, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/about/season/flu-season.htm#anchor_1531424547919
5. 2009 H1N1 pandemic timeline. Centers for Disease Control and Prevention. Reviewed May 8, 2019. Accessed May 23, 2022. https://www.cdc.gov/flu/pandemic-resources/2009-pandemic-timeline.html
6. Trends in number of COVID-19 cases and deaths in the US reported to CDC, by state/territory. Centers for Disease Control and Prevention. Updated June 14, 2022. Accessed June 13, 2022. https://covid.cdc.gov/covid-data-tracker/#trends_dailycases
7. Weekly U.S. influenza surveillance report. Centers for Disease Control and Prevention. Updated May 20, 2022. Accessed May 23, 2022. https://www.cdc.gov/flu/weekly/index.htm
8. Types of influenza viruses. Centers for Disease Control and Prevention. Reviewed November 2, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/about/viruses/types.htm
9. Past pandemics. Centers for Disease Control and Prevention. Reviewed August 10, 2018. Accessed May 23, 2022. https://www.cdc.gov/flu/pandemic-resources/basics/past-pandemics.html
10. Selecting viruses for the seasonal influenza vaccine. Centers for Disease Control and Prevention. Reviewed March 11, 2022. Accessed May 23, 2022. https://www.cdc.gov/flu/prevent/vaccine-selection.htm
11. How flu viruses can change: “drift” and “shift.” Centers for Disease Control and Prevention. Reviewed September 21, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/about/viruses/change.htm
12. Table. Influenza vaccines — United States, 2021-22 influenza season. Centers for Disease Control and Prevention. Reviewed December 10, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/professionals/acip/2021- 2022/acip-table.htm
13. Grohskopf LA, Alyanak E, Ferdinands JM, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices, United States, 2021-22 influenza season. MMWR Recomm Rep. 2021;70(5):1-28. doi:10.15585/ mmwr.rr7005a1
14. How influenza (flu) vaccines are made. Centers for Disease Control and Prevention. Reviewed August 21, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/prevent/how-fluvaccine-made.htm
15. Cell-based flu vaccines. Centers for Disease Control and Prevention. Reviewed November 23, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/prevent/cell-based.htm
16. 171st Vaccines and Related Biological Products Advisory Committee. Summary minutes. FDA. March 3, 2022. Accessed May 23, 2022. https:// www.fda.gov/media/157170/download
17. Frequently asked influenza (flu) questions: 2021-2022 season. Centers for Disease Control and Prevention. Reviewed May 26, 2022. Accessed June 14, 2022. https://www.cdc.gov/flu/season/faq-flu-season-2021-2022.htm
18. Interim US flu vaccine effectiveness (VE) data for 2021-2022. Centers for Disease Control and Prevention. Reviewed March 14, 2022. Accessed May 23, 2022. https://www.cdc.gov/flu/vaccines-work/2021-2022.htm- l?web=1&wdLOR=c896B09AD-CDB2-BD42-A44A-5125D9A4A3C2
19. Past seasons vaccine effectiveness estimates. Centers for Disease Control and Prevention. Reviewed August 26, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/vaccines-work/past-seasons-estimates. html?web=1&wdLOR=cEDA7D328-2317-E142-8419-9BC96655CDC7
20. Diaz Granados CA, Dunning AJ, Kimmel M, et al. Efficacy of high-dose versus standard-dose influenza vaccine in older adults. N Engl J Med. 2014;371(7):635-645. doi:10.1056/NEJMoa1315727
21. Gravenstein S, Davidson HE, Taljaard M, et al. Comparative effectiveness of high-dose versus standard-dose influenza vaccination on numbers of US nursing home residents admitted to hospital: a cluster-randomised trial. Lancet Respir Med. 2017;5(9):738-746. doi:10.1016/S2213-2600(17)30235-7
22. Adjuvanted flu vaccine. Centers for Disease Control and Prevention. September 17, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/ prevent/adjuvant.htm
23. Vaccine effectiveness: how well do flu vaccines work? Centers for Disease Control and Prevention. Reviewed October 25, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/vaccines-work/vaccineeffect.htm#-doe_flu_vax_effect_vary_by_type_subtype
24. CDC director adopts preference for specific flu vaccines for seniors. Media Statement. Centers for Disease Control and Prevention. June 30, 2022. Accessed July 19, 2022. https://www.cdc.gov/media/releases/2022/s0630-seniors-flu.html
25. Flu vaccination coverage, United States, 2020–21 influenza season. Centers for Disease Control and Prevention. October 7, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/fluvaxview/coverage-2021es- timates.htm#:~:text=Vaccination%20coverage%20with%20 %E2%89%A51,points%20from%20the%20prior%20season
26. Protect your patients this flu season. Pharmacist guide and talking points. Centers for Disease Control and Prevention. Updated August 25, 2021. Accessed May 23, 2022. https://www.cdc.gov/flu/pdf/professionals/ vaccination/Flu_HCP_PharmacistTalkingPoints.pdf

Newsletter
Stay informed on drug updates, treatment guidelines, and pharmacy practice trends—subscribe to Pharmacy Times for weekly clinical insights.






