
The efficacy data are consistent with other phase 3 studies, reinforcing lebrikizumab's potential as a first-line treatment across a range of skin tones.

The efficacy data are consistent with other phase 3 studies, reinforcing lebrikizumab's potential as a first-line treatment across a range of skin tones.

The approval comes after positive results from the PhALLCON study, however, further research is needed to confirm immature event-free survival findings.

The oral medication activates an “anti-hunger” molecule which may prevent people from overeating.

To date, this is the only targeted therapy indicated for the treatment of generalized pustular psoriasis.

This approval makes atidarsagene autotemcel the only therapy to be approved in the US for early-symptomatic early juvenile metachromatic leukodystrophy.

Team-based care models improve continuity of care and accessibility for patients.

Charges include conspiracy, mail fraud, and misbranding

The agency will focus on collaboration to protect public health; advancing regulatory approaches; developing standards, guidelines, and best practices; and supporting research that evaluates and monitors AI performance.

Oteseconazole (Vivjoa; Mycovia Pharmaceutical Inc) is the first and only medication for this indication in postmenopausal women and those who do not have reproductive potential.

Pharmacists at this clinic work with patients to improve medication education, affordability, and access to care.

In January 2024, Florida became the only state to allow the importation of medications for public use.

The authors note that understanding the DNA methylation profile in leukemia can help predict whether or not a patient will respond to treatment.

Senators Ron Wyden and Mike Crapo reaffirm their support on 2 health care bills that passed the United States Senate Committee on Finance in 2023.
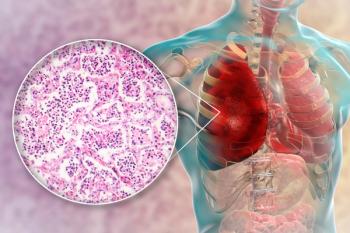
High bacterial load in cerebrospinal fluid was associated with unfavorable outcomes and death for adults with pneumococcal meningitis.

The decision to vote in favor of idecabtagene vicleucel comes after positive phase 3 trial results demonstrating its efficacy compared with standard regimens.

The FDA-approved topical and oral agent is currently indicated for adult patients with patterned alopecia.

Triona Schmelter discusses Opill’s availability, pricing, effectiveness, safety, and impact on addressing barriers to contraceptive access.

Laura Brown, MS, RDN, LDN, discusses the importance of National Nutrition Month, collaborating with pharmacists, promoting sustainable eating habits, and overcoming common nutrition misconceptions

Mila Felder, MD, FACEP, discusses Advocate Aurora Health’s approach to addressing burnout among health care professionals in health care systems.
The disease is complex, but many opportunities exist for pharmacist intervention.

Chimeric antigen receptor T-cell therapies were associated with higher incidences, grades of severity, and longer duration of cytokine release syndrome compared with bispecific antibodies.

Investigators found that 1,1,1 trichloroethane; 2-nitropropane; and 2,4,6 trichlorophenol were significantly associated with asthma symptoms for 3 exposure periods.

In a phase 1/2 clinical trial, lisocabtagene maraleucel helped patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) achieve complete response rates.
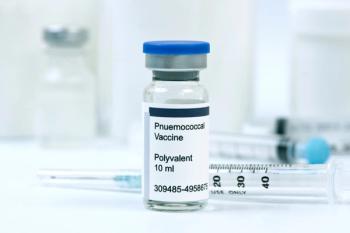
The pneumococcal vaccine was most likely associated with a higher risk of immunological events for those who were immunocompromised

The study showed no clinical differences for remission or asthma at age 28 years based on lung function, body mass index, daily smoking, exposure to parental tobacco smoke, or house dampness .

Their expansive scope of services can ultimately help to elevate the work of the entire community pharmacy.

Compared to placebo, risankizumab significantly reduced symptom severity for patients with moderate to severe disease.

Women who have longer ovarian function and enter menopause later in life tend to live longer, according to women’s health researcher.
The drug is the first FDA-approved agent for treating hepatorenal syndrome.