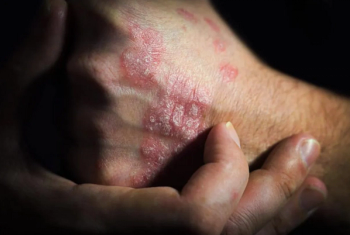
FDA approvals of these biosimilars started in 2016, but only 1 such product has successfully launched.

FDA approvals of these biosimilars started in 2016, but only 1 such product has successfully launched.
This study highlights one benefit of the integrated health system specialty pharmacy model for patients who are prescribed acalabrutinib.