
The second part of a comprehensive review of the specialty pharmaceuticals approved by the FDA in 2017.

The second part of a comprehensive review of the specialty pharmaceuticals approved by the FDA in 2017.

Specialty pharmaceuticals featured prominently in the actions of the FDA in 2017.

A comprehensive review of new oncology molecular entities and previously approved oncology drugs that received expanded indications in 2016, as well as drugs approved in the final days of the year.

Although there were not as many new specialty drug approvals in 2016, there were many already-approved specialty medications that received expanded indications.

The second article in a two-part series examines oncology drug approvals and late-breaking FDA actions.

Part one of a two-part overview recapping significant drugs approved over the last year.

Significant growth has been seen in specialty medication use in long-term care facilities.

New drug approvals include therapies for patients with breast cancer, thyroid cancer, neuroblastoma, and multiple myeloma.

In 2014, the FDA approved notable new therapies for the treatment of cancer, bleeding disorders, inflammatory conditions, cystic fibrosis, pulmonary fibrosis, HIV, hepatitis C, multiple sclerosis, angioedema, and other conditions.

Recently approved drugs for the treatment of cancer include Cyramza, Sylvant, Zykadia, Beleodaq, Zydelig, and Keytruda.

Patient monitoring through a clinical management program allows clinicians to track outcomes and provide outreach, support, and education at key intervals in a patient's therapy.
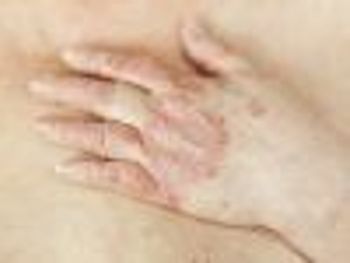
Early diagnosis is the key to successful management of psoriatic arthritis, and treatment of this inflammatory disease is largely dependent on which symptoms are predominant.

Needle phobia can significantly affect the well-being of patients who are serviced by specialty pharmacies, but techniques can be employed to help mitigate its impact.

In 2013, many new medications and new indications were approved.

The year 2013 marked the approval of many new specialty medications, with drug debuts and new indications in areas including oncology, hepatitis C, HIV, multiple sclerosis, and inflammatory disease.

Published: March 13th 2014 | Updated:

Published: February 18th 2014 | Updated:

Published: October 21st 2014 | Updated:

Published: November 16th 2017 | Updated:

Published: January 30th 2018 | Updated:

Published: April 22nd 2014 | Updated: