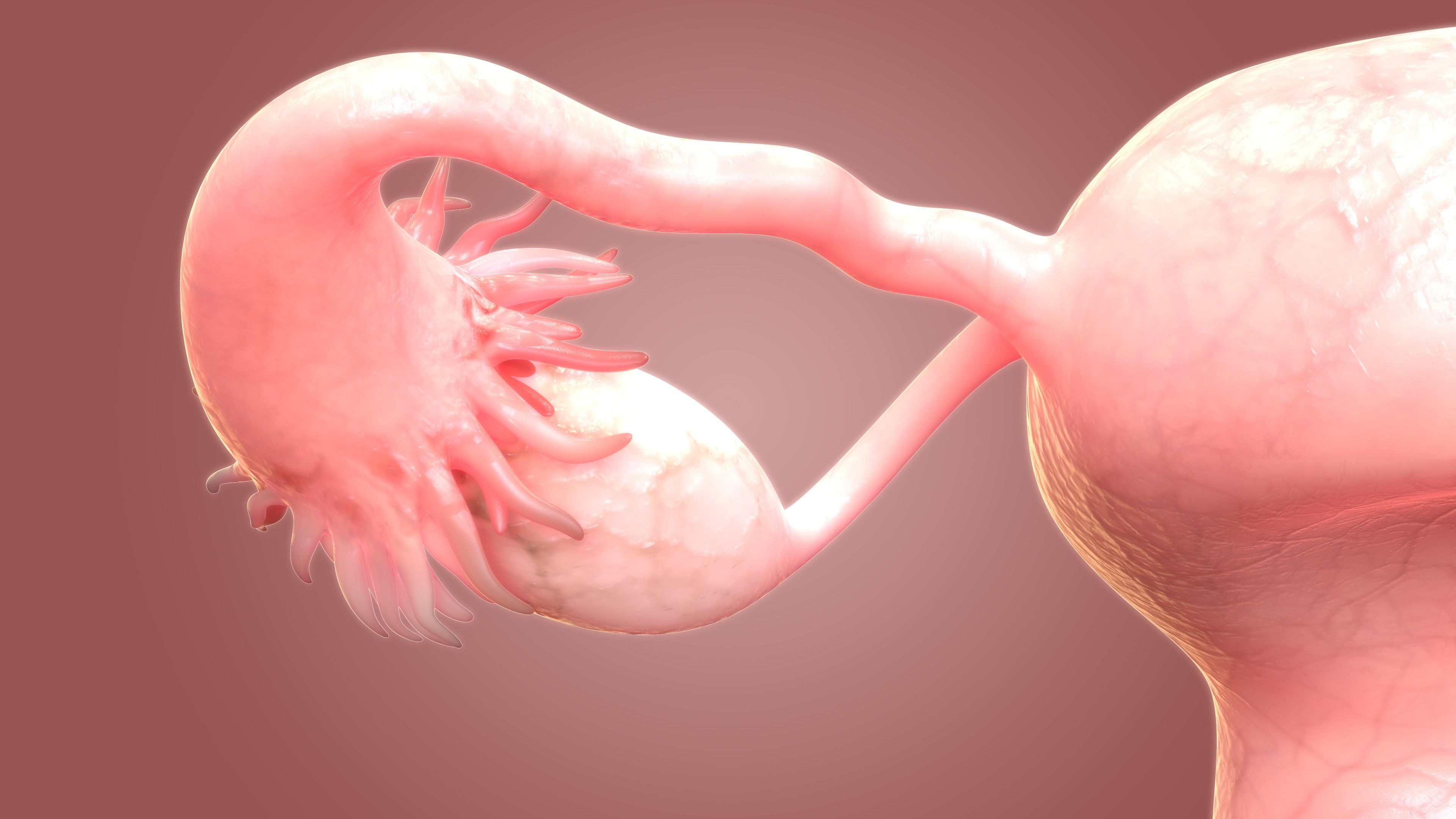
Ovarian Cancer
Latest News

Latest Videos

CME Content
More News

FDA Approves Dabrafenib Plus Trametinib for Unresectable, Metastatic Solid Tumors With BRAF V600E Mutation
In clinical trials, dabrafenib and trametinib showed meaningful efficacy in multiple BRAF-positive tumor types, including in patients with rare cancers who have no other treatment options available.
The ATHENA-MONO trial is investigating the use of rucaparib as front line maintenance treatment in ovarian cancer.
Blinded independent central review analysis also adds confidence to the findings.
Rucaparib at the 400 mg dosage combined with nivolumab and ipilimumab for 4 cycles will continue onto a phase 2 trial for individuals with relapsed ovarian cancer.
The researchers noted that the objective of the study was to assess the trends of 1L PARPi maintenance treatment uptake and PFS of patients with newly diagnosed AOC.
New results from the ATHENA-MONO trial investigating the use of rucaparib in patients with ovarian cancer show promise.

Kirollos Hanna, PharmD, BCPS, BCOP, discussed how PARP inhibitors have changed the treatment landscape of ovarian cancer.

The oral PARP inhibitor is associated with increased PFS, regardless of BRCA mutational status.

Bevacizumab-maly (Almysys; mAbxience) is the third biosimilar of bevacizumab (Avastin) approved in the United States.
Poster at AACR highlights in vitro and in vivosynthetic lethality activity of rucaparib in multiple cancer cell types and PDX tumors that harbor genetic or epigenetic alterations in non-BRCA HRR genes.
Rucaparib (Rubraca) significantly improved progression-free survival when administered as maintenance treatment in newly diagnosed patients with advanced ovarian cancer.

ImmunoGen, Inc submits Biologics License Application under the accelerated approval pathway for mirvetuximab soravtansine monotherapy for patients with FRα-high platinum-resistant ovarian cancer previously treated with 1 to 3 prior systemic treatments.
Niraparib shows promise in patients with newly diagnosed advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer.
Growing evidence indicates that PARP inhibition, both as frontline maintenance and recurrent maintenance, is important for many patients with ovarian cancer.
Only 40% of patients with platinum sensitive disease are receiving a PARP inhibitor in the first-line setting.
Data from the SOLO-1 trial highlight that even with an excellent prognostic group, patients with ovarian cancer still benefit from the use of a PARP inhibitor.
Use of mirvetuximab soravtansine monotherapy in patients with ovarian cancer resulted in meaningful anti-tumor activity, consistent safety, and favorable tolerability.

Black women are at an increased risk of dying from cancer of any type compared to White women, but gynecologic cancers have some of the widest survival gaps.
Hormone replacement therapy can improve quality of life without necessarily increasing the risk of recurrence.

Marina Frimer, MD, FACOG, FACS, gynecologic oncologist at the Northwell Health Cancer Institute, discusses her presentation at the Society of Gynecologic Oncology 2022 annual meeting on the phase 2 trial of maintenance niraparib in patients with stage 3, stage 4, or platinum-sensitive recurrent uterine serous carcinoma.

Thomasina Morris, RPh, MHA, BCOP, pharmacist at Moffitt Cancer Center, discusses what oncology pharmacists should understand about rucaparib when treating ovarian cancer.
Thomasina Morris, RPh, MHA, BCOP, pharmacist at Moffitt Cancer Center, discusses the adverse events patients with ovarian cancer have reported during treatment with rucaparib.

Niraparib is associated with increased progression-free survival, regardless of the presence or absence of BRCA gene mutations.

Trametinib emerged as a novel therapeutic approach for low-grade serous carcinoma due to a high prevalence of activating mutations in the mitogen-activated protein kinase signaling pathway.
Germ cell tumors that occur in places other than the testicles and ovaries are very rare.